Home > > Medical Devices > > Asia Pacific CAUTI Prevention Urology Products Market Size, Trends - 2034
Asia Pacific CAUTI Prevention Urology Products Market - Size, Industry Share, Growth Trends and Forecasts 2025-2034
ID : CBI_3482 | Updated on : | Author : Yogesh K | Category : Medical Devices
Executive Summary
Asia Pacific Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products market is a niche healthcare market dealing with one of the most widespread hospital-acquired infections in the healthcare systems throughout the region. CAUTI contributes to about 30-40 percent of all healthcare-associated infections, and catheter-associated infections contribute 75-85 percent of all healthcare-acquired UTIs, which generates significant clinical and cost-related burdens, necessitating the growth of advanced prevention technology.
The market involves extensive portfolios of the medical devices that are specially designed to reduce the risk of bacterial colonization and infection during urinary catheterization processes. These are antimicrobial coated catheters using silver alloy, nitrofurazone, or antibiotic impregnation technologies that reduce the rate of infection by 40-65 percent over standard catheters, closed drainage systems with anti-reflux valves and needleless sampling ports that prevent retrograde migration of bacteria, catheter stabilization system that lessens urethral trauma and microbial entry points by 30-45 percent, and integrated insertion kits incorporating evidence-based protocols and chlorhexidine-based antiseptics, thereby collectively enhancing patient safety and reducing the incidence of catheter-associated urinary tract infections (CAUTIs) across healthcare settings.
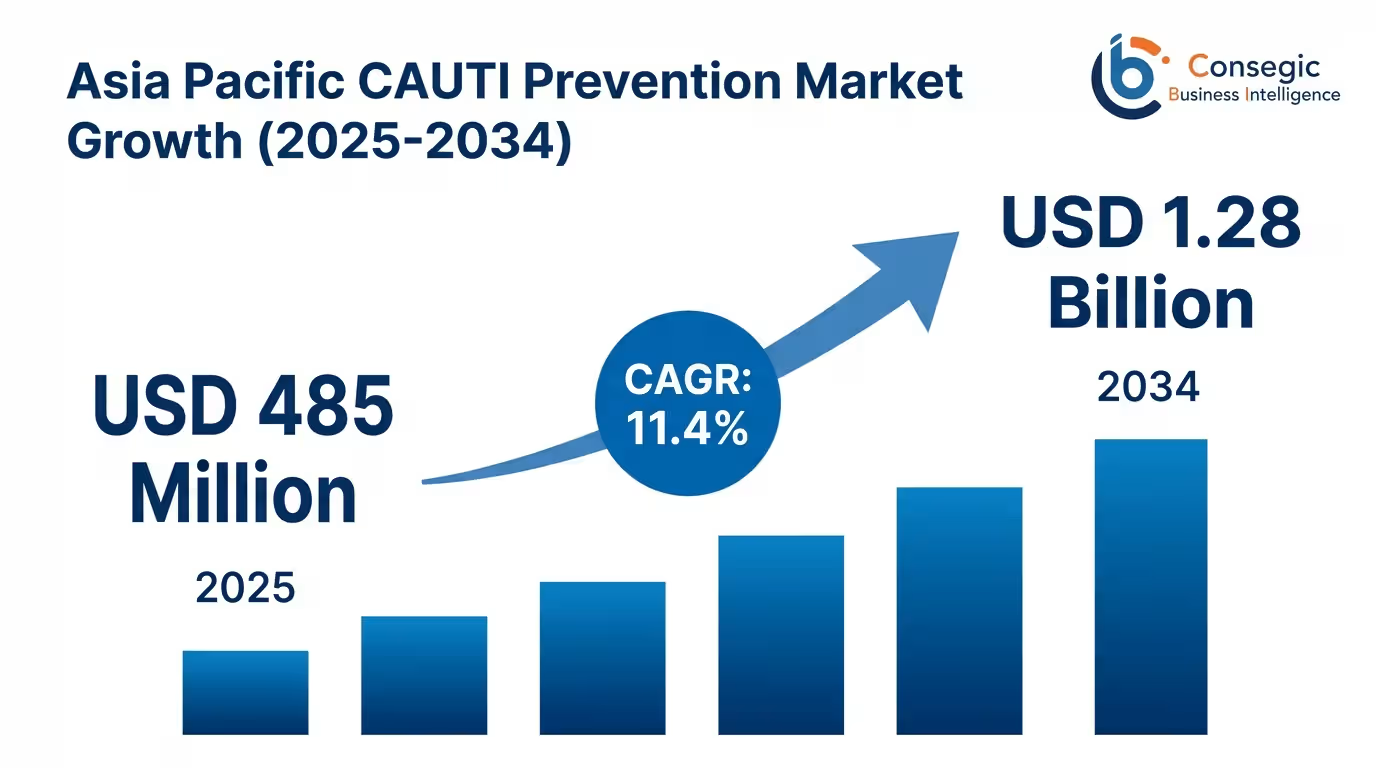
The market has a base year of USD 485 Million in 2025 and a forecast period of 2026-2034 with the value projected to be USD 1.28 Billion in 2034 representing a compound annual growth rate (CAGR) of 11.4% per annum. This strong growth is fueled by the rapid aging populations in which persons aged 65+ represent 420-480 million individuals in the region and which need higher levels of catheterization, the rising awareness of healthcare-associated infections with regulatory guidelines which mandate the reporting of CAUTIs in developed markets, the increasing number of surgical procedures at more than 45–60 million surgeries annually in which perioperative catheterization is a routine procedure, and healthcare quality improvement efforts with zero preventable infections as key performance indicators.
The financial cost of CAUTI is significant, as each instance incurs USD 1,200–3,500 in hospitalization costs due to the necessity of the prolonged length of stay of 2-4 days on average, antibiotic use, and the possibility of complications such as bacteremia at 15-20%. The estimated number of regional CAUTI cases at 800,000-1,200,000 cases per year imply economic cost of USD 1.2-3.6 billion of direct healthcare expenditure, which forms strong value propositions of prevention products despite the premium price of the latter (200-400 percent) as compared to traditional catheters.
Key Market Insights
- Market projected to grow from USD 485 Million (2025) to USD 1.28 Billion (2034) at 11.4% CAGR
- Growth driven by aging population, rising surgeries, and stricter infection control regulations
- High economic burden of CAUTI cases (USD 1.2–3.6 billion annually) boosting demand for prevention solutions
- Market targets CAUTI prevention, a major healthcare-associated infection accounting for 30–40% of HAIs
- India & Southeast Asia are high-growth, price-sensitive markets driven by expanding healthcare infrastructure and medical tourism
Market Scope & Overview
The Asia Pacific CAUTI Prevention Urology Products market includes medical-related devices and consumables that aim to interfere with the pathways of bacterial colonization, the formation of biofilms, and the means of infection transmission related to urinary catheterization. Compared to the traditional urinary catheterization products that are mainly structured around the drainage capabilities, the CAUTI prevention technologies utilize a variety of engineering solutions and material science advances that are specifically aimed at preventing the infection.
Modern antimicrobial catheter technologies exhibit advanced engineering such as silver alloy coating with ionic silver release rates of 0.1-1.0 parts per million in which the antimicrobial activity is sustained between 7-30 days, nitrofurazone-impregnated surfaces with wide-spectrum activity against gram-positive and gram-negative microbes such as multidrug-resistant organisms, antibiotic-eluting surfaces featuring rifampin and minocycline combinations, which greatly decrease adhesion by 80–90%.
Closed drainage systems have several engineering features that stop bacterial entry such as needleless sampling ports that eliminate breach points that result in system breaches in 30-50 percent of traditional systems, one-way anti-reflux valves that stop retrograde urine flow into the collection bag, air vents with anti-microbial 0.2-micron filters that prevent bacterial migration, drainage bag materials with anti-microbial additives that inhibit bacterial growth in collected urine, and emptying mechanisms that prevent contamination in the various disposal processes.
The market has a wide range of clinical uses such as short-term indwelling catheters in surgery and acute care with 1-7 day dwell times of 70-75% of catheterization events, intermediate-term catheters in the critically ill with dwell times of 7–30 days of 15-20% of the total utilization, long-term catheters in chronic conditions and home care with 30 or longer dwell times of 5-10% of the total market volume, and intermittent catheters in patients with neurological conditions, thereby reflecting diverse clinical requirements across acute, critical, and chronic care settings.
Key Market Driver
Increasing Healthcare-Associated Infection Awareness and Regulatory Mandates.
The prevailing structural driving force behind the adoption of CAUTI prevention products is the growing awareness of the effects of healthcare-associated infections on patient outcomes, healthcare expenditure, and quality metrics of the institutions coupled with more demanding regulatory frameworks that require infection surveillance, reporting, and prevention program adoption. The healthcare systems in Asia Pacific record 4–8 million healthcare-associated infections each year, with urinary tract infections among the 30-40% of the total number, generating clinical and financial demands to pursue systematic prevention methods.
In 2018, the Ministry of Health, Labour and Welfare of Japan established mandatory HAI surveillance, to be reported quarterly by 8,500+ acute care hospitals, and public disclosure of the facility-specific infection rates affected the choice of patients and reimbursement decisions. Japanese hospitals with CAUTI rates lower than 2.0 per 1000 catheter-days will receive quality-based payment bonuses of 2-5 percent on applicable diagnostics related group reimbursements, whereas hospitals with higher CAUTI rates than 5.0 per 1000 catheter-days will have payment penalties of 3-8 percent, which constitute USD 15-45 million of payment differences per large hospital system.
The requirements of the urinary catheter management as mandated in Australia under the National Safety and Quality Health Service Standards include infection prevention programs and the daily necessity assessment, documented indication of the insertion, and standard care protocols to follow. The Australian hospitals that are engaged in the national surveillance networks show a median of CAUTI of 1.2-1.8 per 1,000 catheter-days versus 3.5-5.5 per 1,000 catheter-days in hospitals with non-systematic prevention programs which confirm the effectiveness of systematic efforts that include use of special products and procedures.
The National Health Insurance Service of South Korea had introduced value-based purchasing interventions which punish hospitals having excessive rates of HAI including CAUTI, and the economic cost estimated USD 8-25 million per year in large academic hospitals. The Ministry of Health of Singapore has stipulated mandatory reporting and released infection rates per hospital, which put pressure on transparency that led to prevention investment. The regulatory frameworks speed up the move towards the use of antimicrobial catheters, closed drainage systems, and overall prevention bundles, and in regulated markets, the utilization of advanced prevention products in hospitals has increased 45-65 percent higher.
There has been an increased body of clinical evidence supporting CAUTI prevention strategies where systematic reviews and meta-analyses have shown that multimodal interventions that combine products, protocols, and education reduce infection rates by 60–80%, catheter stabilization devices reduce the infection rates by 30-45, closed drainage systems, and comprehensive prevention bundles with antimicrobial catheters have reduced the rate of infections by 25-40 and 40-65 respectively. Economic studies show a 2.5–4.5× return on investment, avoided cost of infection treatment, reduced length of stay and better patient outcome.
Key Market Restraint
Budget Constraints and High Product Costs in Healthcare Systems.
The greatest obstacle to faster uptake of CAUTI prevention products is large cost disparities between advanced antimicrobial product offerings and traditional catheterization products that puts pressure on the budgets of the latter especially given that developing Asia Pacific markets tend to have resource-strained healthcare systems. Antimicrobial-impregnated catheters retail at USD 15–45 per unit versus USD 3-8 for standard silicone or latex catheters at USD 300-500 percent premium prices that translate to USD 1.5–4.5 million in incremental spending for hospitals that undertake 100,000-150,000 catheterizations.
The healthcare systems of emerging markets such as India, Indonesia, Philippines, Vietnam and Thailand have very tight budget constraints with per capita health spending of USD 100–350 per year against USD 4 500-6,500 spent by developed countries with clinical benefits and consequently, they have limited ability to adopt the premium product. The public hospitals in these markets use 65-80 percent in budgets allocated to people budgets, and have little flexibility in medical device purchase with catheter budgets being restricted to USD 3-6 per unit irrespective of infection prevention capabilities.
Critical to the value proposition of CAUTI prevention products are infection baseline rates, treatment costs, and reimbursement structure, which induce market segmentation with adoption being concentrated in high-acuity units with high risks of infection. In ICUs, CAUTI incidences of 4-8 per 1,000 catheter-days exceed general medical-surgical units at 1-3 per 1,000 catheter-days, and thus prevention product investments have a more justified payback period of three to eight months.
Many Asia Pacific markets have a reimbursement structure that does not sufficiently motivate the prevention investments, and the diagnosis-related group payment frameworks of China, Thailand and Malaysia offer fixed payments whether or not an infection occurs, which shifts the financial risk of complications onto healthcare facilities without covering the prevention spending. The lack of clinical evidence in particular regional settings adds complexity to the barriers of adoption, the majority of the antimicrobial catheter efficacy data are based on Western populations, leaving the question of whether the same data would be applicable in the Asia Pacific setting that has different patient demographics and bacterial epidemiology.
Key Market Opportunity
Market Growth in Home Healthcare and Long-Term Care.
The healthcare systems in Asia Pacific have a strong opportunity to expand the use of CAUTI prevention products in high-growth home healthcare and long-term care settings for aging populations with chronic illnesses that need long-term catheterization. In 2024, the population of the region aged 65 and above was 420-480 million, and is expected to increase to over 650-750 million by 2034 with the conditions predisposing to long-term catheterization such as neurogenic bladder of 2-4% of elderly population, urinary retention among 15-25% of men older than 75 and mobility issues resulting in inability to use toilets in 20-30% of long-term care residents.
Home healthcare catheterization has a 5-10% current market volume but with growth rates of 18-25% per year due to healthcare cost containment efforts shifting care out of institutions and into the home, patient preference for aging at home and 75-85% of the elderly preferring home care to an institutional setting where safe catheter management is possible, and advances in technology have made it possible to provide safe catheter management services in non-clinical settings. Japan has 8,000-12,000 home care agencies with a 1.2-1.8 million patients, 180,000–250,000 patients in home care are on long-term indwelling catheter programs or intermittent catheterization.
The long-term care hospitals such as nursing homes, assisted living centers, and rehabilitation hospitals have 3-5 million residents in the Asia Pacific region with a catheterization prevalence of 8-15% translating to 240,000-750,000 catheterized residents at any one point. Such environments record high CAUTI rates of 5-9 cases per 1,000 catheter-days that are attributed to insufficient workforce, inadequate infection control tools, and patient factors such as elderly age and comorbid conditions. The market expansion potential is huge since prevention product adoption in long-term care facilities is 15–25 percent compared to 45–65 percent in acute care hospitals.
Management of neurogenic bladder by intermittent catheterization caters to 2-3 million patients in the region that carry out 4-6 catheterizations per day, and the market is dominated by traditional uncoated catheters priced USD 0.50-1.50 per unit. The premium price of USD 2.50-5.00 per unit for hydrophilic-coated intermittent catheters with 40-60% reduction rates in the rate of urinary tract infection offering manufacturers chances to obtain value based on clinical differentiation and patient outcomes.
Market Segmentation Analysis
By Product Type: Technology and Application Analysis
Antimicrobial-Coated Catheters: Premium Technology Segment
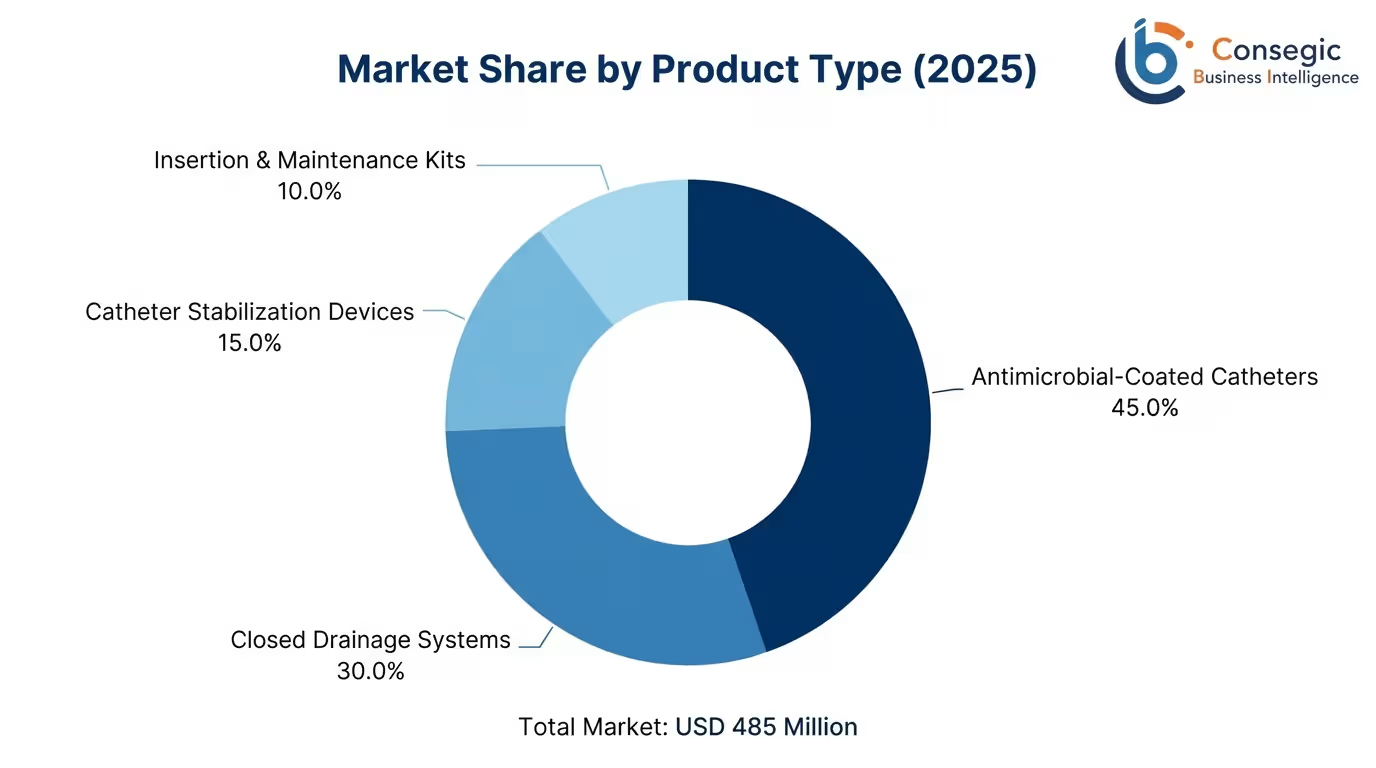
The antimicrobial-coated urinary catheters account for USD 218 million (45.0% of market value) in 2025, and is projected to reach USD 576 million in 2034 at 11.4% CAGR. This category includes silver alloy catheters that use metallic silver forming galvanic reaction to release ionic silver with long-lasting antimicrobial action of 7-30 days, nitrofurazone-coated catheters which show broad-spectrum antibacterial activity especially against gram-negative bacteria such as E. coli causing 60-70% of CAUTI and antibiotic-impregnated catheters based on rifampin and minocycline combinations with evidence of multidrug-resistant organism efficacy.
In meta-analyses involving 15,000-25,000 patients in studies on various randomized controlled clinical trials, clinical evidence has shown that silver alloy catheters decrease CAUTI by 40-50% in short-term catheterization of 1-14 days common in surgical and critical care settings. The cost-effectiveness results reveal the favorable economics in the environment where the baseline infection rates are above 3-4 per 1,000 catheter-days.
Closed Drainage Systems and Collection Bags
The value of closed urinary drainage systems is USD 145 Million (30.0% of market value) with the growth rate of 12.8% CAGR, which includes needleless sampling ports to the system by antimicrobial septa, 1-way anti-reflux valves to prevent retrograde urine flow, antimicrobial filtering vents of 0.2-micron, and drainage bag materials with antimicrobial additives to inhibit bacteria growth. Advanced closed systems show a reduction of CAUTI of 25-40 percent of traditional systems.
Catheter Stabilization and Securement Devices
Catheter stabilization devices are USD 73 Million (15.0% of market value) with 14.5% CAGR as it will stabilize catheter movement in the urethra, which is a major risk factor for microbial entry. Clinical trials prove that stabilization devices can stabilize CAUTI rates by 30-45% by preventing catheter migration and subsequent inflammation that leads to the development of infection vulnerability.
Insertion Kits and Maintenance Products
Catheter insertion kits and maintenance care products account for USD 49 million (10.0% of market value) including sterile insertion trays with chlorhexidine-based antiseptics that enable an 80–95% reduction in perineal bacterial colonization, along with sterile drapes, lubricants, and products that support evidence-based catheter insertion practices.
Regional Market Analysis
Japan: Technology Leadership and Quality-Driven Market
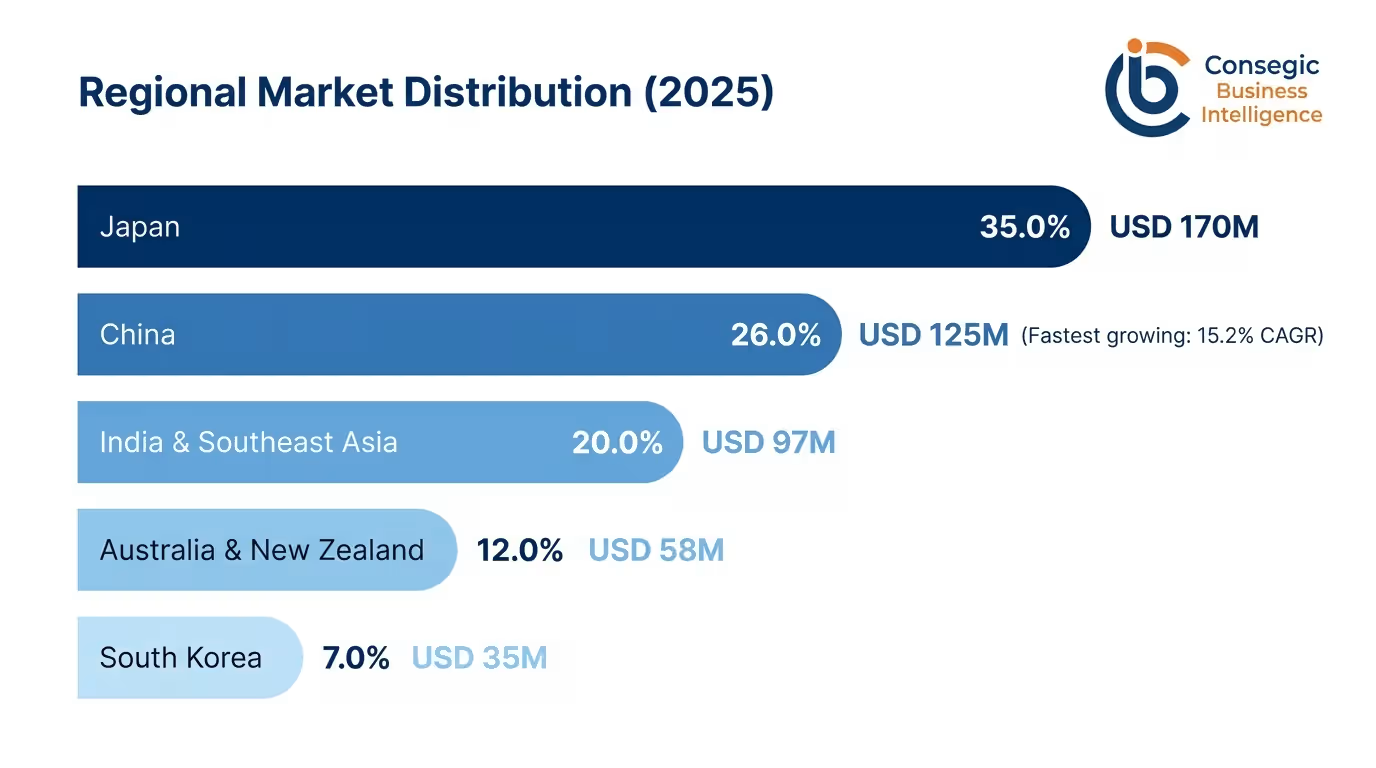
Japan accounts for USD 170 million (35.0% of the overall Asia Pacific market value) in 2025, and USD 448 Million in 2034 and represents outstanding market leadership in terms of advanced regulatory frameworks, quality-based healthcare culture, and extensive infection prevention programs. The nation has 8, 500+ acute care hospitals performing 18-24 million catheterizations per year with 55-65% antimicrobial catheter rates of penetration in acute care units.
Challenges of regulatory environment Regulatory environment involves compulsory HAI surveillance that has been adopted since 2018 that compels quarterly reporting of catheter-associated infections with public disclosure generating transparency pressures and quality competition. Guidelines used in ministry suggest the use of antimicrobial catheters in patients whose catheterization period is likely to take more than 3-5 days or have a high chance of infection. The Japanese hospitals record a median CAUTI of 1.2-1.8 per 1,000 catheter-days making it one of the best in the world in terms of infection prevention performance.
China: Most Volumetric and Fast-Growing Market.
China accounts for USD 125 million (26.0% of market value) with regional growth rate of 15.2% CAGR because of the expansion of the healthcare system, quality improvement efforts, and growing awareness of infections. The nation has 36,000 or more hospitals, 6.8-7.5 million beds, and catheterizations of 85-110 million at antimicrobial catheter penetration rates of 15-25, and 85-110 million in tier-1 and tier-2 city hospitals, respectively.
The recommendation of HAI prevention guidelines 2020 at the national level, which makes the use of antimicrobial catheters when working with high-risk patients and closed drainage systems the standard practice, became effective in the country. The forces in the market are the growth of domestic manufacturing technologies that manufacture antimicrobial catheters at a cost per unit 30-50 per cent less than the imported products, the introduction of government procurement programs that are increasingly specifying features of infection prevention and the growing strength of the private hospital sector that is showing a higher rate of adoption.
Australia and New Zealand: Mature Markets that have Evidence-Based Practice.
Australia and New Zealand comprise USD 58 Million (12.0% of market value) and a growth rate of 9.8% CAGR, and they have a mature healthcare system and a well-established infection surveillance. National Safety and Quality Health Service Standards set out infection prevention programs with certain catheter management conditions. Australian Commission on Safety and Quality runs a national surveillance network which tracks CAUTI rates in 500+ participating hospitals, where median rates are 1.2-1.8 per 1,000 catheter-days.
India and Southeast Asia: Emerging High Growth Markets.
India and Southeast Asian countries are USD 97 Million (20.0% of market value) with growth rate of 13.5% CAGR, growth is driven by healthcare infrastructure development, and increasing quality awareness and expanding middle-class segments. India has 70,000+ hospitals, Southeast Asia has 15,000+ hospitals, and 45-60 million catheterizations with 8-15 catheter penetration by antimicrobial catheters.
The nature of the market is characterized by a large degree of public-private separation whereby the rates of adoption of antimicrobial catheters are 25-40% in private hospitals and 5-12% in public hospitals, which has price sensitivity and necessitates a cost-effective approach, and an increasing trend in medical tourism that uses infection prevention as a competitive factor.
South Korea: Market of the technologically developed country.
South Korea accounts for USD 35 million (7.0% of the market value) with a growth rate of 10.5% CAGR, boasting of technologically developed healthcare system and universal health coverage. The value-based purchasing programs in the National Health Insurance Service punish hospitals in the cases of high HIA rates, which serves to build financial incentives in preventing investments. The implementation of digital surveillance platforms into the comprehensive prevention bundles of Korean hospitals becomes more and more widespread.
Recent Industry Developments
Regulatory Framework Evolution and Quality Mandates (2024-2025)
In 2024, the Ministry of Health, Labour and Welfare in Japan increased the reporting on HAI to the catheter utilisation ratios and calculated numbers (standard rate of infections) to make fair comparisons across facilities. New practices suggest the use of antimicrobial catheters as the preferred choice of catheterization that is likely to more than 3 days, which opens up market space as hospitals can practice in line with their official recommendations.
The National Health Commission of China came up with new infection prevention guidelines with specific requirements of urinary catheters such as the use of closed drainage systems, daily need to assess catheter management with documentation and use of antimicrobial catheters to patients who have poor immunity or in critical care units.
Product Innovation and Technology Advancement (2024-2025).
Top producers came up with new antimicrobial catheters that are next generation and overcome the shortcomings of the old generation products. To deal with the long-term catheterization issues, Becton Dickinson introduced improved silver alloy coating that has prolonged antimicrobial protection of up to 30+ days, compared to up to 7-14 days of traditional silver-coated catheters. It was shown through clinical studies that 55-65% of CAUTI reduction was achieved over standard silicone catheters.
Teleflex Medical launched catheter-free managements of benign prostate hyperplasia to overcome the necessity of catheterization on the elderly male population by permanent implants to disrupt the obstruction and catheterization necessities of 70-80 percent of the treated patients.
Market Expansion and Strategic Partnerships (2024-2025)
With its manufacturing plant in Singapore and yearly production capacity of 25-35 million antimicrobial catheters, Becton Dickson saved on the logistics cost and lead times and developed local supply chain resilience. In China and India, to promote a multifaceted CAUTI prevention program, Medtronic collaborated with major hospital systems and integrated products, clinical guidelines, and digital surveillance solutions in 50-80 hospitals of each country accordingly.
Competitive Landscape and Key Market Players
Strategic Positioning and Market Leadership
Becton Dickson (BD): World Market Leader.
Becton Dickinson is in control of the Asia Pacific market with projected CAUTI prevention product revenues of USD 115-140 Million in 2024, which represent about 23-26% market share of the region, with a complete Bard Medical product portfolio of silver alloy-coated Bardex I.C. and Lubri-Sil I.C. catheters, StatLock catheter stabilization devices, and Infection Control Foley Trays. Competitive advantages are having strong clinical evidence base with 30 plus randomized controlled trials, global manufacturing scale, extensive product line, and well-developed distribution networks.
Teleflex Incorporated: Leader of Innovations.
Teleflex is a strong market player with a reported revenue of USD 85-105 Million which is about 17-20% market share with a focus on product innovation and clinical differentiation. Its competitive advantages are proprietary Rusch silver-coated catheter technology, full closed drainage system portfolio and solid presence in the Arrow and Rusch brands.
Coloplast A/S: Intensive Catheter Specialist.
Coloplast records estimated revenues of USD 65-85 Million, market share of about 13-15% with 40-50% market share of intermittent catheter segment, with hydrophilic coated products. The competitive advantages are SpeediCath product line that has compact designs, massive patient education programs and increasing market share in the long-term catheterization market.
Other Key Market players:
- Medtronic plc - USD 50-70 Million revenues, 10-12 market share.
- Braun Melsungen AG USD 40-60million revenues, presence in Europe and Asia Pacific, good revenues.
- Cook medical - USD 35-55 M revenues, catheter manufacturer (specialty).
- The ConvaTec Group - USD 25-40 Million revenues wound care and urology specialist.
- Hollister Incorporated - USD 20-35 Million revenues, focus on continuance care.
Asia Pacific CAUTI Prevention Urology Products Market Report Insights
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2022–2034 |
| Base Year | 2025 |
| Forecast Period | 2026–2034 |
| Market Size in 2025 | USD 485 Million |
| Market Size in 2034 | USD 1.28 Billion |
| CAGR (2026–2034) | 11.4% |
| By Product Type | Antimicrobial Catheters (45.0%), Closed Drainage Systems (30.0%), Stabilization Devices (15.0%), Insertion/Maintenance Kits (10.0%) |
| By Catheter Type | Indwelling Foley Catheters (70.0%), Intermittent Catheters (20.0%), External/Condom Catheters (7.0%), Suprapubic Catheters (3.0%) |
| By End User | Hospitals (65.0%), Long-Term Care Facilities (18.0%), Home Healthcare (12.0%), Ambulatory Surgery Centers (5.0%) |
| By Region | Japan (35.0%), China (26.0%), India/Southeast Asia (20.0%), Australia/NZ (12.0%), South Korea (7.0%) |
| Key Players | Becton Dickinson, Teleflex, Coloplast, Medtronic, B. Braun, Cook Medical, ConvaTec, Hollister |
| Report Coverage |
|
Key Questions Answered in the Report
What is the present size of Asia Pacific CAUTI Prevention Urology Products market and what is it expected to grow to be? +
In the present market, the Asia Pacific CAUTI Prevention Urology Products market is expected to be USD 485 Million in 2025 and expected to reach USD 1.28 billion by 2034 at a CAGR of 11.4 within the forecast period 2026-2034. This is expanding based on growing patient awareness of healthcare-associated infections, populations that are getting older and hence require more catheterization, surgeries are growing and regulatory requirements mandate CAUTI reporting and prevention initiatives.
Which market shows the best market position and growth potential? +
Japan is 35.0 percent of the total value of the Asia Pacific market which exhibits market leadership in terms of sophisticated regulatory systems, quality-based healthcare culture and penetration of antimicrobial catheters in acute care facilities at 55-65 percent. China has the highest growth at 15.2% CAGR due to expansion of healthcare system with 36,000+ hospitals, quality improvement efforts, and government procurement programs are increasingly defining infection prevention features.
What are the major clinical and economic gains of CAUTI prevention product? +
Clinical trials show a reduction in the CAUTI rate of 40-65% with antimicrobial-impregnated catheters, 40-50% reduced with silver alloy catheters, and 50-65% reduced with antibiotic impregnated catheters. Every CAUTI imposes the financial burden of USD 1,200 to 3,500 in the hospitalization costs in terms of the prolonged hospital stay, antibiotic therapy and complications. Prevention products yield returns of 2.5-4.5 times product expenditure in terms of treated cost avoidance and better performance.
What are the major regulatory frameworks of the market adoption? +
The mandatory HAI surveillance in Japan instituted in 2018 mandates reporting CAUTI quarterly with public disclosure and quality-based payment bonuses to hospitals with a rate that is lower than 2.0 per 1,000 catheter-days and penalties to those with higher than 5.0 per 1,000 catheter-days. The National Safety and Quality Health Service Standards in Australia provide full-fledged infection prevention programs with specifications of catheter management. The value-based purchasing programs in South Korea impose monetary penalties to hospitals with high rates of HAI that have an annual financial implication of USD 8-25 million.
Who are the strongest manufacturers, what is their market standing? +
Becton Dickinson, Teleflex, Coloplast and Medtronic are market leaders, with market shares of 23-26, 17-20, 13-15, and 10-12 respectively, due to comprehensive Bard Medical product offerings including silver alloy-coated catheters and stabilization devices, Rusch silver-coated technology and closed drainage systems, intermittent catheter business, and diversified portfolio of medical devices, respectively.
What are the opportunities within the home healthcare and long-term care settings? +
Homecare catheterization is 5-10 percent of existing market volume that is growing by 18-25 percent/year due to cost containing measures and patients wanting to age at home. The residents at LTC facilities are 3-5 million with 8-15% catheterization prevalence showing high CAUTI rates of 5-9 per 1,000 catheter-days and low confidence in prevention products of 15-25% against 45-65% in acute care hospitals, with a high market expansion opportunity.
What are the contributions of closed drainage systems to the prevention of CAUTI? +
Engineering features such as needleless sampling ports contributing to the elimination of breach points that lead to the impairment of system integrity, one-way anti-reflux valves that prevent retrograde bacterial migration, antimicrobial filters in the air vents (0.2-micron) that block the passage of bacteria, as well as antimicrobial-additive drainage bag materials decrease CAUTI rates by 25-40 percent in 30-50 percent of traditional systems.
What are the significant cost impediments to market adoption? +
The antimicrobial-impregnated catheters will demand USD 15-45/unit versus USD 3-8/unit on standard catheters, which amounts to a 300-500 premium price, translated into USD 1.5-4.5 million of incremental expenditure/years to hospitals that perform 100, 000 -150,000 catheterizations annually. The healthcare systems of the emerging markets with a USD 100-350 annual per capita health expenditure have very limited budget constraints, with public hospitals, spending 65-80% of the budgets on personnel, leaving no flexibility to be in a position to purchase premium products.
What is the mechanism of action of catheter stabilization devices in the prevention of infections? +
Catheter stabilization devices decrease CAUTI rates by 30-45% by preventing catheter mobility that leads to trauma of the urethra, access points of microbes, and patient discomfort. Clinical research proves that catheter migration and movement induce inflammation and micro-trauma that offer avenues of bacteria entry, and stabilization keeps this risk factors stable at regular positions compared to the highest growth percentage of 14.5% CAGR.
What do you see as the future trends and opportunities in technologies over the next 15 years (2034)? +
Great possibilities are revealed in alternative antimicrobial development such as copper-based coating and bacteriophage-impregnated material, integration of digital health with smart catheters with sensors tracking infection biomarkers, catheter-free management technologies that will remove catheterization requirement in 70-80% of patients with benign prostatic hyperplasia, expansion of intermittent catheterization that will serve 2-3 million patients with hydrophilic-coated products with 40-60% infection reduction, and comprehensive prevention bundles combining products and protocols, thereby driving next-generation innovation and significantly reducing CAUTI incidence across care settings.