Home > > Pharmaceuticals > > Pharmaceutical Membrane Filtration Market Size & Outlook - 2035
Pharmaceutical Membrane Filtration Market - Size, Share, Industry Trends, and Forecasts (2025 - 2035)
ID : CBI_3501 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
Why is Membrane Filtration Important in Pharmaceutical Manufacturing?
Pharmaceutical membrane filtration involves the use of filtration techniques for the separation of particles, microorganisms, and impurities, which are used for the production of pharmaceuticals. The use of filtration techniques is common in the production of drugs, water treatment, and the production of biologicals. The filtration techniques used are classified as follows: microfiltration, ultrafiltration, nanofiltration, and reverse osmosis.
The market growth is attributed to the increasing demand for biopharmaceuticals and the production of sterile drugs. Rising regulatory requirements for product safety and quality are supporting filtration adoption. Advancements in membrane materials and filtration technologies are improving process efficiency and supporting market expansion.
Key Highlights
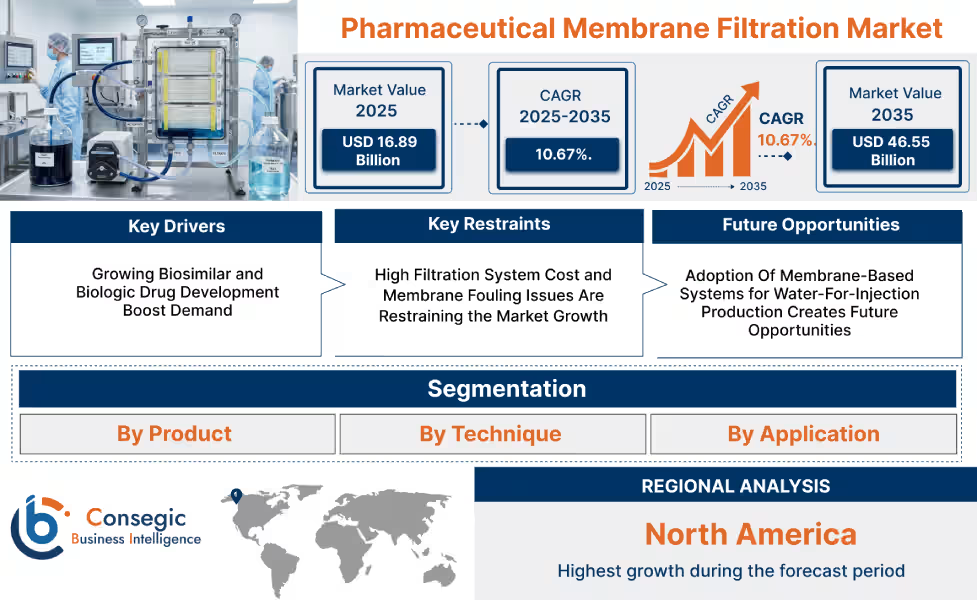
- The market is projected to grow from USD 16.89 billion in 2025 to USD 46.55 billion by 2035, at a CAGR of 10.67%.
- Rising biologics and biosimilars development is driving filtration demand.
- Increasing chronic disease burden is supporting pharmaceutical production.
- Membrane systems for water-for-injection are creating growth opportunities.
- Stringent sterility regulations are accelerating filtration adoption.
- Asia Pacific dominates, while North America shows the fastest growth.
- Key players include Merck KGaA, Danaher Corporation, Sartorius AG, 3M Company, and Thermo Fisher Scientific Inc.
Drivers
Growing Biosimilar and Biologic Drug Development Boost Demand
The pharmaceutical membrane filtration market is expanding due to the growth of biologic and biosimilar drug development. Biologic drugs are complex molecules obtained from living organisms. The manufacturing of biologic drugs involves the use of advanced purification techniques and sterile membrane filtration technologies. Membrane filtration is commonly used during the production of biologic drugs to remove host cell contaminants, virus contaminants, and impurities.
For instance, according to the U.S. Biosimilar Market Entry Challenges and Facilitating Factors Final Report (2025)" published by the United States Department of Health and Human Services, "The FDA had approved 63 biosimilars of 18 reference biologic products by the end of 2024.
Therefore, the growth of biosimilar and biologic drug production is creating a need for pharmaceutical membrane filtration technologies.
Increasing Prevalence of Chronic Diseases
The incidence of chronic diseases, including cancer, diabetes, and cardiovascular diseases, is rising, which in turn increases the need to produce drugs on a large scale. Pharmaceutical companies are increasing the production of drugs to cater to the treatment requirements of these diseases, which in turn increases the need for membrane filtration technologies. The need to produce drugs on a large scale increases the use of microfiltration and ultrafiltration technologies in the upstream and downstream processes. For example, World Health Organization, chronic diseases are likely to cause 90 million deaths annually by the year 2048, which translates to 86% of the total deaths if the current trend continues.
CMO Expansion Increases Filtration Outsourcing
The expansion of CMOs results in the outsourcing of drug manufacturing activities for pharmaceutical and biopharmaceutical companies. Outsourcing of production activities demands flexible and scalable filtration systems to accommodate different product pipelines. CMOs invest heavily in advanced membrane filtration technologies to ensure high throughput and meet strict regulations for different client projects. For instance, CPHI estimates that by 2028, 40% of global capacity for biologics production is expected to be handled by CMOs, while 45% of this capacity is expected to be located in Asia. This trend is expected to promote the use of single-use and modular filtration systems for increased operational efficiency and reduced turn-around time.
Challenges
High Filtration System Cost and Membrane Fouling Issues Are Restraining the Market Growth
Pharmaceutical membrane filtration systems need specialized membranes, filtration units, and monitoring equipment. Pharmaceutical membrane filtration systems are costly in terms of initial investment and maintenance. Smaller pharmaceutical manufacturers may face difficulty adopting advanced filtration technologies. Thus, the high system costs involved and the fouling of the membranes are factors that restrain the growth of the pharmaceutical membrane filtration market.
Opportunities
Adoption Of Membrane-Based Systems for Water-For-Injection Production Creates Future Opportunities
The pharmaceutical industry is increasingly employing advanced membrane filtration technologies to enhance efficiency in the processes involved. Water for Injection (WFI) is an essential component that is utilized for the manufacture of injectable pharmaceutical products. The conventional method of producing WFI is distillation. For instance, in June 2024, Asahi Kasei has commenced the sale of a membrane system for the manufacture of Water for Injection (WFI) utilizing the Microza hollow fiber membranes. This is an alternative to the distillation method while ensuring the efficiency of the system is maximized. Thus, it is, therefore, expected that the application of membrane-based WFI production systems will boost the efficiency of the pharmaceutical manufacturing process.
Biosimilars & Advanced Therapies Boost Filtration Need Creates Opportunity
The increased investment in biosimilars and advanced therapy technologies, cell therapy, and gene therapy requires high precision in filtration technologies. The need for precise control over contaminants and the nature of the biological molecule is significant in the production of cell therapy and gene therapy. The membrane filtration system plays an important role in the critical separation and purification steps in the production of biologics. The increased pipeline of complex biologics requires specialized filtration technologies with high selectivity and performance, hence contributing to the growth of the market. In January 2026, Toray Industries, Inc., developed a new membrane filtration technology with improved separation efficiency and lower energy consumption.
Global Regulatory Landscape for Pharmaceutical Filtration Technologies:
The regulatory guidelines for pharmaceutical membrane filtration are based on the compliance standards for pharmaceuticals in terms of sterility, validation, and good manufacturing practices. The pharmaceutical industry in the US is based on guidelines set by the U.S. Food and Drug Administration, which includes validated processes for drug filtration and integrity testing. The pharmaceutical industry in Europe is based on guidelines set by the European Medicines Agency, which includes good manufacturing practice Annex 1 guidelines for the manufacture of sterile products.
Regulatory bodies such as Central Drugs Standard Control Organization in Asia Pacific countries are enhancing their standards in pharmaceutical production and water for injection systems. These regulations ensure product safety and quality, thereby increasing the use of advanced membrane filtration technology in pharmaceutical production.
Pharmaceutical Membrane Filtration Market Segmental Analysis:
By Product
The market includes membrane filters, filtration systems, and accessories.
Dominant Segment
Membrane Filters
Largest revenue share in 2024. High use in microorganism removal supports demand. Strong need for sterile drug production. High growth rate due to biologics and vaccines.
Fastest Growing
Filtration Systems
Fastest CAGR during the forecast period. Integration with automated equipment improves efficiency. Demand for large-scale production supports growth. Advancements in automation accelerate adoption.
By Technique
The market includes microfiltration, ultrafiltration, nanofiltration, and reverse osmosis.
Dominant Segment
Microfiltration
Largest revenue share in 2024. High efficiency in bacterial removal fuels the need. Strong use in pharmaceutical drug production fuels adoption. Strong need for safe drug production fuels the need.
Fastest Growing
Ultrafiltration
Fastest CAGR during the forecast period. Strong use in protein and biomolecule separation fuels growth. High growth rate due to biologics drug production. High growth rate due to need for advanced purification technologies.
By Application
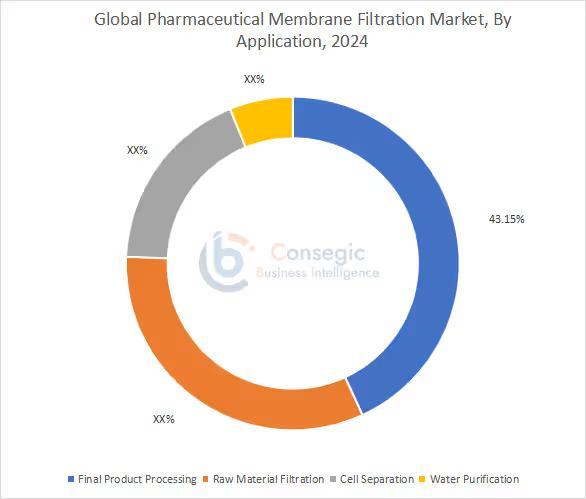
The market includes final product processing, raw material filtration, cell separation, and water purification.
Dominant Segment
Final Product Processing
Largest revenue share of 43.15% in 2024. Ensures removal of contaminants and safe drug production. Strong focus on drug sterility fuels the need. High growth rate due to vaccines and injectables.
Fastest Growing
Water Purification
Highest CAGR is expected to be achieved in the forecast period. High demand for pure water in the production process also drives the market. Membrane systems ensure high purity standards. Strict guidelines for water-for-injection accelerate adoption.
Geographic Breakdown:
The regions include North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America in the market.
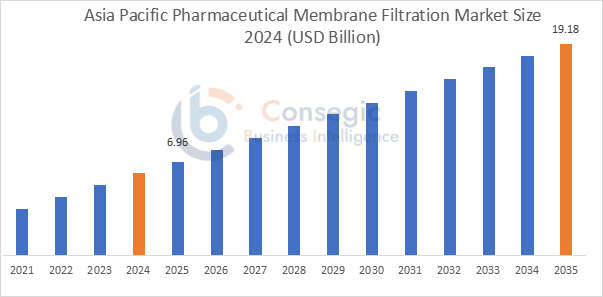
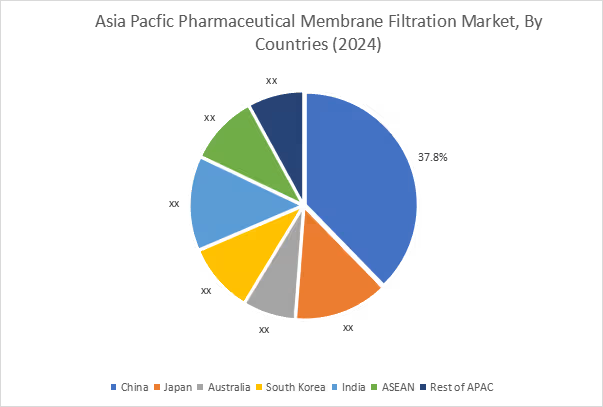
Asia Pacific held the largest market share of 41.20% in 2025, with a value of USD 6.96 billion, and is projected to reach USD 19.18 billion by 2035. China dominated the market in the region, holding the highest market share, 37.80%, in 2025. The Asia Pacific market is driven by the growth in pharmaceutical production, production of biologics, and increased investment in the production of vaccines and drugs in China, India, Japan, and South Korea. For instance, the Economic Survey 2025-26 stated that pharmaceutical sector is moving to high-value products such as biosimilars and complex generics, while the country continues to provide 20 percent of the world's generic medicines and export them to 191 countries.
North America is expected to witness the highest growth rate in the forecast period. The growth in the region is attributed to high production levels in the biologics market, increased investment in pharmaceutical R&D and manufacturing, and stringent regulatory guidelines for the production of sterile drugs in the US and Canadian pharmaceutical industries. For instance, in June 2025, Cytiva announced to increase its filtration manufacturing capacity in Pensacola, Florida, by 20% to cater to the increased demand from the biopharmaceutical market.
Major Companies Operating in the Pharmaceutical Membrane Filtration Industry
The pharmaceutical membrane filtration industry is a competitive one, with major players in the life sciences and bioprocessing providing cutting-edge technology to the pharmaceutical and biotechnology industry across the globe. The industry major players are focusing on R&D, new product development, acquisition, and capacity expansion to strengthen their positions.
Major companies operating in the pharmaceutical membrane filtration market include:
- Merck KGaA(Germany)
- Danaher Corporation(U.S.)
- Sartorius AG(Germany)
- 3M Company (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- GE HealthCare Technologies Inc. (U.S.)
- Asahi Kasei Corporation (Japan)
- Repligen Corporation (U.S.)
- Meissner Filtration Products, Inc. (U.S.)
- Eaton Corporation plc (Ireland)
Recent Industry Developments:
- In September 2025, Thermo Fisher Scientific Inc. acquired the Purification & Filtration division of Solventum Corporation for 4 billion USD. This acquisition enhances its portfolio in the areas of filtration and membrane technologies, especially for the pharmaceutical and bioprocessing industries.
- In June 2025, UCB has announced its plan to expand operations in the US, including the construction of a new biologics manufacturing plant, which will enhance the production capacity of advanced biologic medicines.
Pharmaceutical Membrane Filtration Market Report Insights:
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2019-2035 |
| Market Size in 2035 (USD Billion) | USD 46.55 Billion |
| CAGR (2025-2035) | 10.67% |
| By Product |
|
| By Technique |
|
| By Application |
|
| By Region |
|
| Key Players |
|
| Report Coverage |
|
Key Questions Answered in the Report
How big is the pharmaceutical membrane filtration market? +
The pharmaceutical membrane filtration market size is estimated to reach over USD 46.55 Billion by 2035 from a value of USD 15.26 Billion in 2024 and is projected to grow by USD 16.89 Billion in 2025, growing at a CAGR of 10.67% from 2025 to 2035.
Which segmentation details are covered in the pharmaceutical membrane filtration report? +
The pharmaceutical membrane filtration report includes specific segmentation details for product, technique, application, and regions.
Which is the fastest segment anticipated to impact the market growth? +
In the pharmaceutical membrane filtration market, the water purification segment is anticipated to be the fastest-growing segment during the forecast period due to the rising demand for high-purity water in drug production.
Who are the major players in the pharmaceutical membrane filtration market? +
The key participants in the pharmaceutical membrane filtration market are Merck KGaA (Germany), Danaher Corporation (U.S.), Sartorius AG (Germany), 3M Company (U.S.), Thermo Fisher Scientific Inc. (U.S.), GE HealthCare Technologies Inc. (U.S.), Asahi Kasei Corporation (Japan), Repligen Corporation (U.S.), Meissner Filtration Products, Inc. (U.S.), Eaton Corporation plc (Ireland), and others.
What are the key trends in the pharmaceutical membrane filtration market? +
The pharmaceutical membrane filtration market is shaped by several key trends including increasing demand for sterile filtration in biologics production and rising adoption of single-use filtration systems.