Home > > Pharmaceuticals > > Asia Pacific Fetal Bovine Serum Market Size, Demand & Analysis - 2034
Asia Pacific Fetal Bovine Serum Market - Size, Industry Share, Growth Trends and Forecasts (2025-2034)
ID : CBI_3459 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
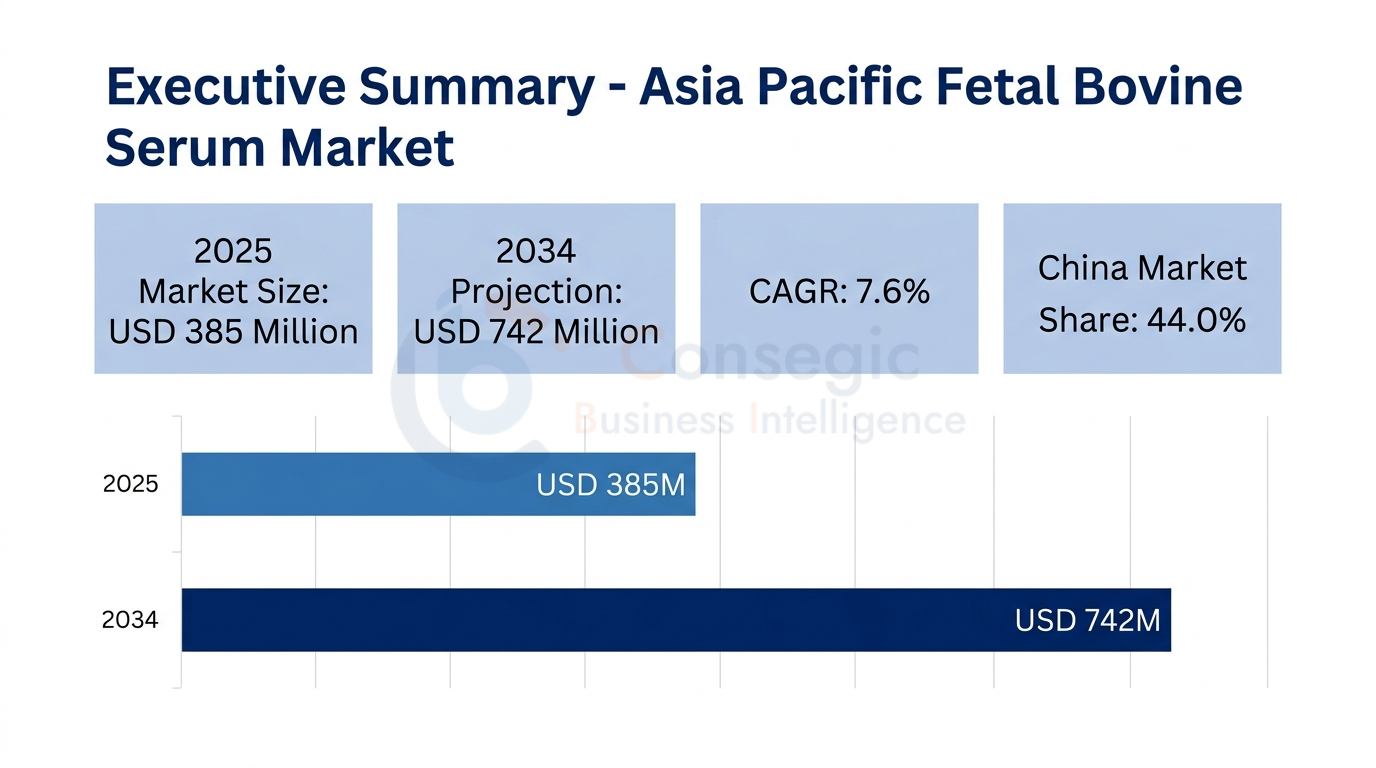
Asia Pacific Fetal Bovine Serum Market Executive Summary
The Asia Pacific fetal bovine serum (FBS) market plays a critical role in supporting biopharmaceutical manufacturing, academic research, vaccine production, and regenerative medicine across the region. Academic research, vaccine production and regenerative medicine industries in the area, which are growing rapidly. FBS is a vital cell culture supplement that provides growth factors, hormones, vitamins, and attachment factors and as such, it is indispensable in the mammalian cell proliferation of drug discovery study, biologic manufacturing, stem cell research, and clinical diagnostics applications in a variety of industry verticals.
The Asia Pacific FBS market is estimated to grow from USD 385 million in 2025 to USD 742 million by 2034, at a CAGR of 7.6% during 2025–2034 which has a strong Compound Annual Growth Rate (CAGR) of 7.6 percent within the 2026-2034 forecast period. This is a rapid growth curve that far surpasses the average growth rates in other global markets as a result of the strategic relocation of biomanufacturing facilities to Asia, government-funded biosimilar efforts to estimate USD 25 billion and expanding biosimilar manufacturing capacity, particularly in China, and the rise of contract research and manufacturing organizations to projects with the global pharmaceutical pipelines.
Market Dynamics
Key Driver
Biopharmaceutical Production and Cell Therapeutics.
The primary driver of demand is of Asia Pacific FBS is the shift of the region into an international biopharmaceutical production centre due to cost-saving factors, access to human resources, and more sophisticated regulatory and licensing regulations. The biologics market in China stood at USD 58 billion in 2024, with a growth rate of 18.2 percent per year; monoclonal antibodies, recombinant proteins, and cell-based therapies are some of the biologics products that demand massive use of FBS in research and development and also in the commercial-scale manufacturing processes.
The biosimilar production facilities in India, South Korea and China generate large FBS consumption patterns. Also there is the biosimilar industry of India worth USD 5.8 billion in 2024 which is run by the existing manufacturers of Biocon, Dr. Reddy’s Laboratories and Cipla, with the combined bioreactor capacity to serve global supply chains. The average number of liters of FBS used in the cell banking process, seed train expansion, and optimization of production phases per 10,000-liter bioreactor production campaign of mammalian cell-based products is 800-1,500 liters.
The most rapidly expanding section of application is regenerative medicine and cell therapy development, and there are dramatic regulatory developments throughout the region. The approval of seven products based on cell therapy by the regulatory authorities in Japan, the four CAR-T therapies approved by China and the advanced therapy medicinal products framework in South Korea have enabled the ability to produce cell therapies at commercial scale. Clinical use of Mesenchymal stem cells takes 15-25 liters of FBS to produce 100 billion cells and cell therapies and clinical trials are becoming more active in regions (34 percent of 2023-2024).
Biotechnology efforts by governments give a significant push in various nations. The 14 th Five-Year Plan of China has earmarked RMB 180 billion (USD 25 billion) to be used in the development of biotechnology, paying special attention to the biopharmaceutical manufacturing capacity and local innovation. The National Biotechnology Development Strategy of India is aimed at reaching USD 150 billion value of the biotechnology industry by the year 2025, in a manner that will aid in the growth of infrastructure that will directly lead to higher consumption of FBS in terms of research and manufacturing applications.
55 the world had invested USD 685 Million in the development and commercialization of 55 technology, with technology suppliers estimated to spend around 9.2% of the revenue on system applications to enhance the efficiency of gasification and syngas cleaning system, development of advanced feedstock preprocessing technologies, and system integration with downstream uses. There is an essential change in the industry towards integrated concept of biorefinery including generation of power, biofuel production coupled with biochemical synthesis with massive adoption in the industrial, utility-scale, and distributed energy uses.
Key Market Restraint
Ethical Concerns and Supply Chain Vulnerabilities.
Regardless of the strong growth drivers, the market has major structural limitations in terms of ethical concerns with regard to the mode of collection of FBS and structural weaknesses in the supply chain. The FBS collection procedure, which entails cardiac puncture of bovine fetuses that are procured at slaughter plants, is facing increasing scrutiny from animal welfare groups and regulatory authorities that emphasize the 3Rs principle (Replacement, Reduction, Refinement), and institutional review boards, which are adopting more stringent procurement guidelines.
To ensure these concerns are addressed, major pharma corporations have developed corporate policies requiring new biologics programs to develop serum-free processes. In 2024, about 42 percent of the surveyed biopharmaceutical firms said they were currently undertaking initiatives to eliminate FBS in their upstream manufacturing operations due to mitigation of supply chain risks, concerns about batch-to-batch fluctuations, and corporate sustainability obligations. The trend of serum-free manufacturing process preferred by the European Medicines Agency impacts on the world development strategies and therefore affects Asian manufacturers in Asia Pacific which sell to the world market.
The vulnerabilities of the supply chain lead to continuous uncertainty in the market, and the impact of the COVID-19 pandemic revealed the essential reliance on foreign suppliers. Shipment delays, exportation, and slaughterhouses led to the 40-60 percent rise in prices in 2020-2021, and periodic biosecurity threats such as foot-and-mouth disease outbreaks in source countries have the potential to effectively impede exports over the long term. The main sources of imports in Asia Pacific such as Australia and New Zealand are challenged by the issue of climate variability that impacts cattle populations, as well as strict biosecurity measures that may interfere with the continuity of the supply.
The distribution of quality specifications into the regional markets complicates and increases the cost involved in managing the supply chain. Various nations have different importation standards, test procedures and documentation requirements, causing challenges to the efficient distribution, and raise the overall cost of ownership to end-users operating in multiple jurisdictions.
Key Market Opportunity
Domestic Production Competencies and Higher Product Differentiation.
The strategic market expansion opportunity focuses on building regional FBS production facilities to decrease the reliance on imports and create value with top products differentiation. The present capacity in the region is only sufficient to cater to 15-20 percent of the demand in Asia Pacific, which offers massive prospects to localize the supply chain and source domestically on a guaranteed quality basis.
China has embarked on large-scale domestic FBS production by government-assisted projects that set up collection facilities in the Inner Mongolia, Qinghai, and Xinjiang provinces that have a population of cattle exceeding 100 million head. In 2024, the China National Biotec Group introduced a 420 million (USD 58 million) FBS production plant in Hohhot, with an annual production capacity of 180,000 liters and quality parameters to satisfy the international standards of pharmaceutical products and reduce the reliance on imports.
The large cattle stock of about 300 million animals in India constitutes a large untapped potential of production but religious and cultural inhibitions on slaughtering cows, in addition to disjointed slaughterhouses infrastructure and cold chain logistics make commercial development of FBS production difficult. Intercontinental FBS manufacturing companies and Indian biotechnology firms are taking strategic alliances with an eye to investigating ethics-based models of collection and end-to-end quality assurance systems that do not ignore cultural factors but satisfy the demands of the industry.
The high-end product differentiation will provide a significant value capture in a variety of niche segments. There are ultra-low IgG FBS variants with applications in developing hybridoma and monoclonal antibody which fetch 60-80 percent price premiums compared with standard formulations. High-value niche segments with low production potential in the region are charcoal-stripped FBS used in hormone-sensitive cell culture preparations, gamma-irradiated variants used to increase viral inactivation, and stem cell-qualified lots with proven pluripotency maintenance properties.
Market Segmentation Analysis
By Product Grade
Premium and GMP-Grade FBS (38.0% of market value) is the most rapidly expanding and most valuable segment, serving biopharmaceutical production, clinical-grade cell therapy product production, and vaccine creation, which must have extensive quality records. This type consists of serum that passes stringent specifications of viral screening panels of 18-20 pathogens, endotoxin levels of less than 10 EU/mL (as compared to 50 EU/mL of research grade), hemoglobin content of less than 20 mg/dL, and mycoplasma testing in a variety of detection methods with batch-to-batch consistency validation in 15-20 cell lines.
Research Grade FBS (42.0% of market value) is used in academic institutions, contract research organization and preliminary research in pharmaceuticals where cost and quality considerations are balanced. This product category incorporates standard FBS meeting minimum sterility standards, mycoplasma testing standards, and basic viral screening specifications, where endotoxin values are less than 50 EU/mL and hemoglobin levels are less than 25 mg/dL, which presents reliable performance at affordable costs in the daily cell culture workload.
Specialty FBS products (20.0% of market value) are ultra-low IgG forms to use in hybridoma assays where minimal interference with immunoglobulins is needed, dialyzed FBS to be used in metabolic and hormone research, charcoal-stripped FBS to be used in endocrinology experiments, heat-inactivated preparations to use in complement-sensitive cell lines and stem cell-qualified lots to be used in pluripotency maintenance. These are niche products with special technical needs that would warrant 50-120 percent higher prices than their standard counterparts.
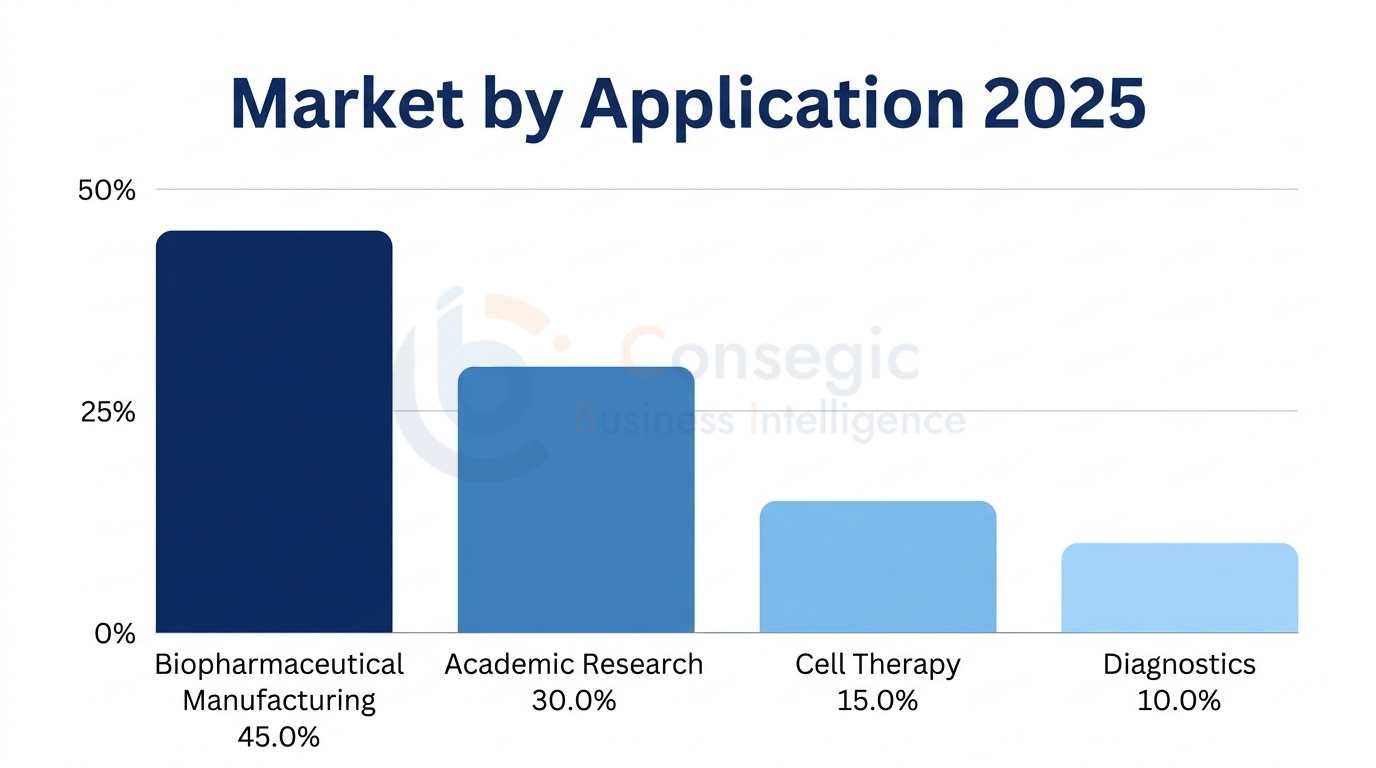
By Application
The consumption patterns are dominated by the Biopharmaceutical Manufacturing (45.0% of market value) that comprises of monoclonal antibody production, Recombinant protein manufacturing, vaccine development and biosimilar manufacturing in the growing manufacturing structure of the region. Chinese biopharmaceutical firms, such as WuXi Biologics, Genor Biopharma, and Innovent Biologics, have a combined bioreactor of over 800000 liters with extensive FBS use in the process development, cell line development, master cell banking operations, and scale-up processes.
Academic and Research Institutions (30.0% of market value) are a long-term demand by university, government research institutes and non-profit-making organizations engaged in basic cell biology research, drug discovery screening, and translational medicine research. In 2024, China spent USD 468 billion on research, and 18 percent of total R&D investment was on life sciences, whereas in India the Department of Biotechnology budget allocation amounted to USD 580 million that will be used on stem cell research, vaccine development, and translational medicine programs.
Cell Therapy and Regenerative Medicine (15.0% of market value) becomes the fastest-growing application segment due to clinical developments through mesenchymal stem cell therapies, CAR-T cell production as well as tissue engineering uses. In 2020, there were 147 regional cell therapy clinical trials, and in 2024, these expanded to 312, which necessitated a high rate of cell expansion protocols, manufacturing procedures development, and regulatory validation trials, which required significant cell consumption of FBS.
Diagnostics and Other Applications (10.0% of market value) encompass in vitro diagnostic reagent production, viral vaccine production using cell culture systems and quality control testing applications that need FBS as a key component of the media to support various analytical and manufacturing processes.
Through End-User: Customer Segmentation and Purchasing Patterns.
The largest category of customer (50.0% of market value) comprises of Pharmaceutical and Biotechnology Companies, the consumption patterns of which are high-volume purchases, high quality standards, and long-term supply agreements that are less focused on pricing and more on reliability and regulatory compliance. Such organizations attach importance to capabilities of suppliers such as detailed documentation, assistance services, security of supply chains, and not only the cost.
Contract Research and Manufacturing Organizations (25.0% of market value) such as CROs and CDMOs with global pharmaceutical clients need a source that is flexible, offers a variety of products and can create products quicker to meet the needs of changing project schedules and client-specific product demands. The need to meet the growing demands of large players such as WuXi AppTec, Syngene International, and Samsung Biologics results in the creation of high demand due to their growing service offerings.
There is the case of the Academic and Government Research Institutes (20.0% of market value) which indicate price sensitivity harmonized with quality requirements in that the purchasing decisions are based on the institutional procurement policies, budget limitations on the research grants and the need of the research program in the long term. This segment usually uses research grade products at a standard of quality that allows it to be published and allow regulatory submission.
Diagnostic Companies and Others (5.0% of market value) entails in vitro diagnostic companies, veterinary pharmaceutical companies and industrial biotechnology applications that have special demands on individual features of quality attributes and regulatory standards of quality compliance.
Regional Market Analysis
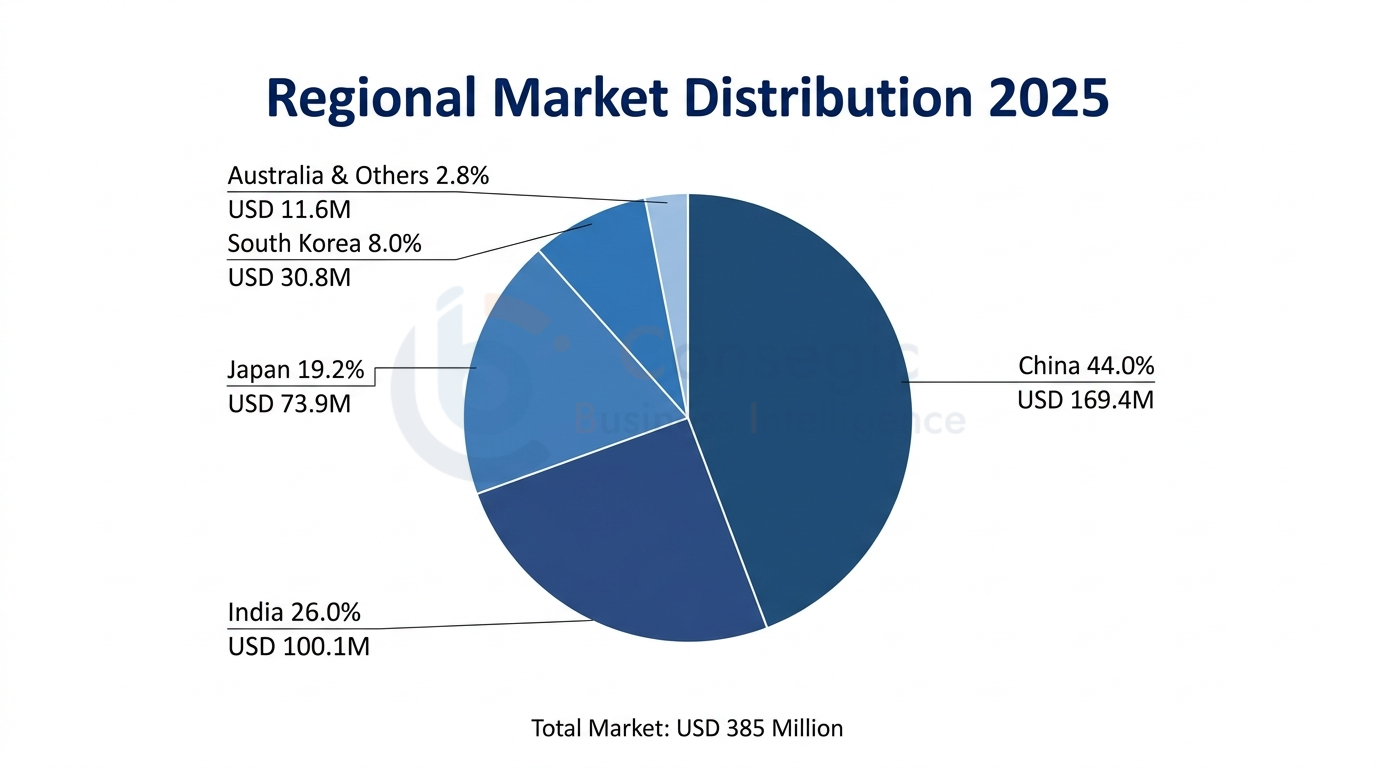
Biggest Market: China - Infrastructure and Research Investment in manufacturing.
The USD 169.4 Million (44.0% of market value) of China in 2025 will increase by USD 333.9 Million by the year 2034 with a CAGR of 7.8 percent. Chinese market is under the advantage of the second-largest pharmaceutical market in the world with USD 175 billion, robust expansion in the biopharmaceutical manufacturing capacity that is part of the Made in China 2025 initiative and the extensive government programs aimed at the development of biotechnology under the 14th Five-Year Plan.
Infrastructure of biopharmaceutical manufacturing includes more than 180 commercial scale biologics facilities run by such companies as WuXi Biologics (12 facilities and a bioreactor capacity of 580,000 liters), Shanghai Henlius Biotech, an Innovent Biologics and BeiGene. In 2024, the National Medical Products Administration granted 76 new biologics, including 23 monoclonal antibodies and 4 cell therapies, which sustained the FBS requirement across the development and manufacturing lifecycles.
The use of academic research is also high, as China has 3,200 universities and research institutions that have around 2.1 million employees working in the life sciences areas. Chinese Academy of Sciences has 104 institutes of research with the total life sciences research spending of more than USD 8.5 billion annually to finance the comprehensive cell culture research work in the fields of basic biology, drug discovery, and translational medicine use.
The domestic FBS production programs are meant to lower the amount of imports that is currently pegged at 85 percent of total consumption. Hohhot plant and production operations in Inner Mongolia of the China National Biotec Group were geared towards the combined production volumes of 250,000 liters by 2026 but the technical and commercial constraints are continuous due to the quality validation, international certification demands and scale-up difficulties.
Rapidly Expanding Market: India - Biosimilar Production and Research Development.
India symbolizes USD 100.1 Million (26.0% of market value) in 2025, which is expected to increase to USD 208.3 Million in 2034 with the greatest regional CAGR of 8.5 percent. The Indian market is exhibiting a tremendous growth potential owing to the leadership in biosimilar manufacturing, the rising contract research and manufacturing strengths, the broad government biotechnology development programs and rising domestic pharmaceutical research and development.
India, which has 60 percent of the global market share by volume, is a part of the biosimilar industry which has 47 commercial-scale biologics plants where companies such as Biocon, Dr. Reddy's Laboratories, Cipla, and Lupin produce products which are shipped to more than 120 countries. The total bioreactor capacity of the industry is in excess of 1.2 million liters and high consumption of FBS in cell line development, process optimization, regulatory validation research and commercial manufacturing processes.
The global pharmaceutical companies such as Syngene International, Anthem Biosciences, Piramal Pharma Solutions and Aurigene Discovery Technologies are contract research and manufacturing companies with annual revenues over USD 2.8 billion each in 2024. These companies need different FBS product portfolios to serve different client needs, capability to source flexibly based on particular projects, and quality assurance systems based on international regulatory standards.
The infrastructure of the academic research consists of 45 new biotechnology research centers developed in 2020-2024 as the part of the National Biotechnology Development Strategy, and the government research funding grows 24 percent annually. The budgetary allocation by the Department of Biotechnology promotes stem cell research programs, vaccines development programs, and translational medicine research that needs significant consumption of FBS in various researches.
Developed Markets: Japan, South Korea and Australia - High Quality Orientation.
The characteristics of a mature market with a focus on high quality products, complete regulatory compliance and gradual adoption of serum-free alternatives can be shown in Japan (USD 73.9 Million, 19.2% of market value). The intensive regulatory system of the Pharmaceuticals and Medical Devices Agency and the high level of regenerative medicine in Japan, with seven approved products of cell therapies by 2024, support a long-term demand in the production of ultra-high-quality FBS variants in accordance with the most challenging requirements.
The growth of South Korea (USD 30.8 Million, 8.0% of market value) is strong due to the expansion of biopharmaceutical manufacturing and Samsung Biologics has the largest single-site biologics manufacturing facility in the world that has the capacity of 620,000 liters. The framework of the Advanced Regenerative Medicine and Advanced Biopharmaceuticals Act of the country contributes to the rapid development of cell therapy, and 23 clinical trials were launched in 2024, which require unique FBS formulations.
Australia is both a major regional supplier and a major consumer with domestic FBS production of over 4,00,000 liters a year on both established collection networks and local consumption of about USD 11.6 Million (3.0 percent of market value) in the support of academic research institutions and other growing biopharmaceutical development efforts.
Competitive Landscape and Key Market Players
Global Leadership: Thermo Fisher Scientific - End to End Product Portfolio and Supply Security.
Thermo Fisher Scientific has dominated the Asia Pacific market with an estimated USD 108-120 million regional revenues in the year 2024, which constitute about 28-31 percent of market shares of the products of the FBS in all its categories. Its competitive positioning is based on extensive product portfolios of research grade-GMP certified formulations under the industry-leading Gibco brand, wide coverage of quality documentation to support regulatory filings, technical support infrastructure that includes regional application scientists and integrated supply chain management that ensures product availability during market shocks.
The quality control measures at the company involve triple 0.1-micron filtration, full viral screening of 18-20 pathogens, endotoxin levels by validated LAL tests, hemoglobin quantification, and validation of the company on 15-20 cell lines, which have been widely used. Strategic programs involve increasing regional distribution channels, setting up technical support centres in Shanghai, Bangalore and Singapore and coming up with special products in new applications such as cell therapy production and 3D cell culture systems.
The benefits associated with supply chains are availability of global sourcing networks across Australia, New Zealand and the North America region so that the supply chain can be secured in the event of disruptions in the region, and the provision of end-to-end cold chain logistics infrastructure that maintains the integrity of the products throughout the collection to final delivery to the end-users. The One Shot aliquot packaging and overall origin-traceability certification are the industry standards of quality assurance and regulatory standards in the company.
Valmet continues to dominate the market with a projected USD 485 Million 55 revenue in 2024 amounting to 9.8 per cent global market share. Its business unit, the Energy and Environmental business, is an entity that deals with full gasification systems and integrated biorefinery.
Quality Specialization: Sigma-Aldrich (Merck KGaA) - Technical Excellence and Premium Positioning.
Sigma-Aldrich as a subsidiary of Merck KGaA has a good Asia Pacific presence with the projected 2024 revenues of USD 65-73 million, which is about 17-19 percent market share with its specific strength in premium and specialty FBS markets. The competitive differentiation approach focuses on ultra-low IgG preparations to use in hybridoma applications, fully defined FBS with complete certificate of analysis records and specialty preparations such as dialyzed, charcoal-stripped and gamma-irradiated versions used in niche research markets.
The quality assurance measures have involved high-order testing methodology such as fully developed mycoplasma detection by a number of validation techniques, broad biochemical characterization and batch-to-batch consistency validation by growth promotion testing in varied cell lines. Premium products have price premiums of 60-90 percent supported by high level performance features, detailed technical documentation and specialized technical support services.
The regional strategy of the company is to provide high-value customers that need specialized formulations and extensive technical assistance such as pharmaceutical companies developing complex biologics, academic research institutions carrying out the latest cell biology research, and contract research organizations with clients with high-quality demands all over the world.
New Competition and Regional Competitors.
Cytiva (Danaher Corporation) looks to its robust bioprocessing equipment base to package FBS solutions with single-use bioreactor systems and chromatography packages and carve out value in terms of workflow solutions that are of specific interest to contract development and manufacturing organizations. The HyClone brand offers economically competitive options without compromising the quality of what is suitable in an industrial requirement.
Biowest has established a presence in the Asia Pacific by forming strategic alliances with distributors in the region, providing attractive prices on research-grade quality FBS and at the same time meeting European Union quality standards and full traceability reports that are attractive to low-end academic as well as research clientele.
Regional experts such as Bovogen Biologicals (Australia) take advantage of the location to deliver premium products with short delivery times and nonstandard quantities ordered, and the local distributors in China and India offer lower-cost options and bypass complicated importation processes to academic clients with tight budgets.
The Industry Latest Developments.
Capacity Expansion and Localization of Supply Chain to Domestic (2024-2025)
In March 2024, China National Biotec Group contracted a modern FBS production plant in Hohhot, Inner Mongolia, which will become RMB 420 million (USD 58 million) of infrastructure that can manufacture 180,000 liters per year. The facility also has automated collection facilities, full cold chain logistics facility, and advanced quality control laboratories with state of the art analytical instrumentation to do viral screening, endotoxin quantification and biochemical characterization tests to the international pharmaceutical standards.
The strategic positioning in Inner Mongolia gives access to cattle population of over 24 million head and animal welfare protocols, which are intended to meet the international standards and eliminate ethical issues. First batches in production were subjected to intense validation testing with quality requirements of being equivalent to imported high-grade FBS of established Australian and New Zealand suppliers.
The response of large global suppliers is to increase regional distribution capacity and cold-storing retailing. In late 2024, Sartorius increased its media and reagents logistics center in South Korea to better serve the Northeast Asian markets, and Merck KGaA invested in the expansion of its Mumbai distribution center, more cold storage capacity to stock biologics during demand peaks.
Technology Integration and Quality Assurance Improvement (2024-2025)
Top FBS providers launched blockchain-traceable systems in 2024, which allowed substantial tracking of the product up to the source recovery plants all the way to processing, testing, and distribution channels. The implementation of blockchain in Thermo Fisher scientific covers both Australian and New Zealand collection networks and offers customers with clear documentations of animal sourcing, collection process, processing parameters and quality tests findings in support of regulatory submissions and audit criteria.
The use of advanced quality control technologies has been established throughout production facilities, such as automated viral screening platforms, real-time endotoxin monitoring and analytical capabilities which would provide uniform product quality and batch-to-batch reproducibility. These are technological advances that meet the needs of the pharmaceutical industry of validated, traceable biological materials as well as supply chain risk management processes.
Strategy Partnerships and Serum-Free technology Development (2024-2025).
The large biopharmaceutical producers increased the use of serum-free media by entering into strategic alliances with specialty media vendors to consider the long-term sustainability issues and supply chain risks. In June 2024, WuXi Biologics partnered with Sartorius to create chemically-defined media formulations to support the CHO cell-based manufacturing processes, aiming to achieve total removal of FBS in commercial-scale biologics manufacturing processes in 36 months without loss of product quality properties.
In 2024, Samsung Biologics embarked on a wholesale program of converting its entire process of client manufacturing to serum-free media formulations, transforming 40 percent of their manufacturing processes. The project is inclusive of comprehensive process development research, comparability checks, and regulatory documentation of changes in manufacturing with maintenance of product quality and regulatory adherence.
Market Consolidation and Acquisition Activity (2023-2024)
Competitive environment went through consolidation as large participants in the market bought regional distributors and specialty suppliers to consolidate market positions and win supply chain access. In November 2023, Sartorius purchased a majority share of a biological reagent distributor located in South Korea, BioSolution, worth USD 42 million, giving it direct market access and technical support in the Korean biopharmaceutical industry.
In March 2024, Merck KGaA acquired Australian FBS processor BioResources which granted direct access to raw material sourcing and processing capabilities and diversified premium product portfolios in Asia Pacific markets. These strategic acquisitions also indicate the industry trends that focus on vertical integration and improvement of supply chain security.
Asia Pacific Fetal Bovine Serum Market Report Insights
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2022–2034 |
| Base Year | 2025 |
| Forecast Period | 2026–2034 |
| Market Size in 2025 | USD 385 Million |
| Market Size in 2034 | USD 742 Million |
| CAGR (2026–2034) | 7.6% |
| By Product Grade | Research Grade (42.0%), Premium/GMP Grade (38.0%), Specialty Products (20.0%) |
| By Application | Biopharmaceutical Manufacturing (45.0%), Academic Research (30.0%), Cell Therapy (15.0%), Diagnostics (10.0%) |
| By End-User | Pharma & Biotech (50.0%), CROs/CMOs (25.0%), Academic Institutes (20.0%), Others (5.0%) |
| By Origin | Australia/New Zealand (65.0%), North America (20.0%), South America (10.0%), Asia (5.0%) |
| By Region | China (44.0%), India (26.0%), Japan (19.2%), South Korea (8.0%), Others (2.8%) |
| Key Players | Thermo Fisher Scientific, Merck KGaA, Cytiva, Biowest, PAN-Biotech, Bovogen, Capricorn Scientific |
| Report Coverage |
|
Key Questions Answered in the Report
What is the size of Asia Pacific fetal bovine serum market? +
The 2025 Asia Pacific FBS market size is USD 385 Million which includes research grade, premium grade GMP grade and specialty FBS products applied in biopharmaceutical manufacturing, academic and cell therapies development, vaccine production and diagnostic applications all over the region.
How fast is the Asia Pacific FBS market likely to grow? +
Market growth rates are predicted to rise by 7.6 percent between 2026 and 2034 to USD 742 Million, which is highly above the world market averages because there is a consistent biopharmaceutical manufacturing growth, government-sponsored biotechnology research, and cell-based research activities are increasing.
What is the biggest Asian Pacific market? +
The largest market share of 44.0 percent is held by China (USD 169.4 Million in 2025) due to the high presence of biopharmaceutical manufacturing infrastructure, with more than 180 facilities of commercial-scale biologics, extensive governmental biotechnology investment initiatives totaling USD 25 billion, and high consumption of academic research in the 3,200 plus universities and research institutes.
Which market has the best growth prospects? +
India has the most promising growth trend of 8.5 percent per capita; it is driven by the biosimilar manufacturing leadership of 60 percent of the worldwide market share in volumes, the developing contract research and manufacturing capacities which supply to clients worldwide, and government biotechnology development programs of 24 percent annual budget.