Home > > Pharmaceuticals > > Clinical Trial Management System Market Size & Forecast 2035
Clinical Trial Management System (CTMS) Market Size By Delivery Mode (Cloud-Based, On-Premise), By Component (Software Solutions, Implementation & Professional Services), By End-User (Pharmaceutical and Biotechnology Companies, Contract Research Organizations), by Region and Forecast 2035
ID : CBI_3431 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
Clinical Trial Management System (CTMS) Market Scope & Overview:
The global clinical trial management system (CTMS) market is a revolutionary segment to the healthcare information technology and pharmaceutical research infrastructure industry as mission-critical software platforms that are the central databases managing patient recruitment, site selection, regulatory compliance, budgeting, and operations workflow of the multiplex, multi-country drug development programs. In 2024, the market was valued at USD 1,850 Million followed by USD 1,980 Million in 2025 with a projection of enormous growth to USD 4,285 Million in 2035. This growth profile reflects a forecast of compound annual growth rate (CAGR) of 8.0 per cent in the 2026-2035 forecast period caused by pipeline acceleration, rising complexity and globalization of clinical trials, and adherence to regulatory compliance, rising use of decentralized and hybrid trial designs, and digital transformation efforts in the pharmaceutical and biotechnology industries.
Regional Trends and Insights
North America’s CTMS Market | USD 871 million in 2025 (44% of the global market) | Projected to reach USD 1,885 million by 2035 at an 8% CAGR
Europe’s CTMS Market | USD 614 million in 2025 (31% of the global market) | Projected to reach USD 1,328 million by 2035 at an 8% CAGR

Clinical trial management systems are operational backbones of clinical research with integrated systems managing key operational areas such as site feasibility analysis, patient recruitment and retention tracking, investigator and site management, regulatory document management, financial management and invoicing, study milestone tracking and real-time reporting dashboard. Worldwide, there are about 455,000 registered clinical trials in 195 countries in 2024, with pharmaceutical and biotechnology firms spending over USD 185,000 Million annually on research and developing initiatives, of which about 60-65% of total research and development activities is clinical trials, which pose considerable pressure on finding efficient trial management technologies that can help to shorten the cycle time, enhance the quality of data collection, and more effectively utilize the resources.
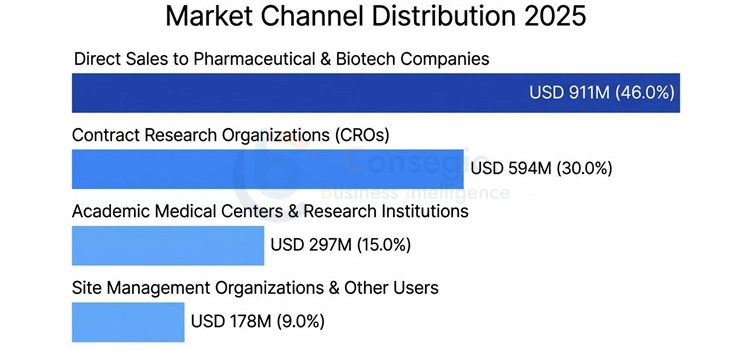
The Distribution Analysis of Market Channels is as follows:
The market structure of the clinical trial management system offers four different sales and delivery channels with differentiated value propositions and implementation models targeting various categories of customers. The overall market revenue breakdown will show a very high variation based on the type of customer, the size of the organization and geographical aspects:
- Direct Sales to Pharmaceutical and Biotech Companies USD 911 Million (46.0% of total market value)
- Contract Research Organizations (CROs): USD 594 Million (30.0% market revenue)
- Academic Medical Centers and Research Institutions: USD 297 Million (15.0% of market share)
- Site Management Organizations and other users USD 178 Million (9.0 percent of market value)
In 2024, the investments in the development of CTMS technology, cloud infrastructure, artificial intelligence integration, and interoperability improvement globally reached USD 285 Million, with software vendors accounting about 14.2% of their revenues. A paradigm shift in the industry is toward integrated clinical trial platforms comprising CTMS, EDC, randomization and trial supply management (RTSM), electronic trial master file (eTMF), safety reporting systems, and massive switching to such systems is underway among large pharmaceutical companies, global CROs, and emerging biotechnology companies.
Dynamics in Clinical trial management system market (DRO).
Key Drivers:
Raising the Clinical Trials Complexity and Globalization.
The greatest structural impetus is increasing complexity of contemporary clinical trials with growing numbers of patients, complicated endpoints, adaptive clinical trials, precision medicine methods and multi-site implementation across different regulatory jurisdictions. The Phase III average trial currently has around 760 patients in 40-60 sites in 12-18 countries, which is 185 percent more geographically dispersed than trials that were run in 2010, generating significant operational complexity that demands complex management systems.
The complexity of the clinical trial protocol has grown exponentially with the average protocol length standing at 230 pages (a 175 procedure per patient 12-18 months to enroll patients), and the protocols must include 175 procedures, a 20-fold increase over the past decade, in contrast to the former 95 pages, 105 procedures per patient, and 8-10 months to enroll patients. It is this complexity that leads to 42 percent increased screen failure rates, 35 percent longer cycle times and significantly high cost of operations averaging USD 2.6 billion to introduce a new drug to the market and this poses a dire need to seek technological solutions that make processing of drugs simplistic and more productive.
Move Towards Decentralized and Hybrid Clinical Trials.
The growing momentum of the adoption of decentralized clinical trial (DCT) models that contemplate the use of remote patient monitoring, home visits, telemedicine sessions, and direct-to-patient drug shipment has placed considerable strain on CTMS platforms that have been able to support geographically distributed trial activities, coordinate mobile health providers, and integrate the results of various digital health technologies. The COVID-19 pandemic increased DCT adoption by about 5-7 years with 78% of pharmaceutical companies currently using decentralized components of trial designs versus 32% before the pandemic.
Current CTMS systems are developing to organize site and home health nurse visits, monitor remote visits and telehealth visits, distribute distributed vendors (home nursing, courier, labs, imaging), and facilitate risk-based monitoring (RBM) and central management. A positive impact of sponsors adopting CTMS heavily co-located with eCOA/ePRO and eConsent services has been noted to result in 20-30 less site monitoring visits, 12-20 less protocol deviation rate, and enhanced patient retention by 5-10 percentage points in long-term studies.
Data integrity and regulatory Compliance Requirements.
Strict regulatory demands by FDA, EMA, PMDA, and other health authorities worldwide insist on a lot of documentation, audit trail, data integrity measures and quality management systems during the execution of clinical trials. FDA 21 CFR Part 11 standards of electronic records and electronic signatures, ICH-GCP standards of good clinical practice, and developing data privacy rules such as GDPR and HIPAA, present complicated compliance environments that necessitate certified software systems that have strong security, access control and audit features.
Key Restraints:
Expensive in Implementation and resource needing.
Enterprise CTMS implementations incur large capital investment of USD 850,000-4,500,000 in large pharmaceutical businesses basing on the number of users, customization needs, complexity of integration, and model of deployment (cloud/ on-premise). Such expenses include software licensing, implementation services, data migration, system validation, user training and maintenance which pose big impediments on small biotechnology companies and academic research institutions with small budgets.
Enterprise-wide deployments usually take 12-24 months to implement, and would need project-specific teams, rich IT capabilities, process reengineering, and change management initiatives. Organizations note averages in the costs of implementation of USD 1,850-3,200 user plus software, services, and internal costs, and annual cost of maintenance is 18-22% of the original license charges.
System Integration Issues and Data Pods.
Operational activities of clinical trials entail the integration of various distinct systems such as EDC systems and databases, safety networks, laboratory information management systems (LIMS), images, eTMF repositories, as well as finance. Making these systems flow together with no repetition of data entry in these systems technically becomes difficult, and 68 percent of the organizations have indicated integration challenges to be the main challenges during implementation.
Future Opportunities:
CTMS with AIs and Predictive Control.
This integration of CTMS platforms with artificial intelligence, machine learning, and sophisticated analytics represents innovative possibilities to predictive site selection, optimized patient recruitment, risk-driven monitoring, and automated protocol deviation detection. With an accuracy of 85-92, AI powered CTMS systems use historical trial data, site performance metrics, patient demographics, and real-world evidence to predict enrollment rates and pinpoint high performing sites as well as to optimize resource allocation.
According to reports by early adopters of AI-enhanced CTMS, it has shown reductions in overall timelines (12-18) in terms of total trials in mid-size Phase II/III portfolios, and fewer physical visits to sites (15-25) to conduct monitoring, and enhancement of site-at-risk identification (20-30) before major delays set in.
Integrated Clinical Platform Convergence.
The trend of converting clinical trial platforms into a single vendor system with CTMS, EDC, RTSM, eTMF, safety reporting, and patient engagement features open up significant possibilities to vendors providing end-to-end solutions. Unified platforms remove complexity of integration, lower the total cost of ownership 28-42% than best-of-breed strategies, and shorten implementation schedules by 35-50% using pre-integrated modules and standardized workflows.
Market Segmentation Analysis
By Delivery Mode: Deployment Model Analysis
Cloud-Based CTMS: Dominant Growth Driver
Cloud based CTMS have commanding market leadership of USD 1,386 Million or 70.0% of total market value in 2025 which is projected at USD 3,342 Million in 2035 with the fastest growing market segment of 9.2% CAGR. The models of cloud deployment have interesting benefits such as reduced initial capital needs, shorter implementation cycles of 4-8 months compared to on-premise systems of 12-24 months, automatic software updates, scalability to satisfy changing user numbers, and availability that facilitates decentralized trial models and international cooperation.
On-Premise CTMS: Legacy and Specialized Applications
On-premise CTMS systems are estimated to be USD 594 Million (30.0) of total market value, which is offered to organizations with certain data sovereignty demands, broad customization needs, or substantial investment in legacy infrastructure. On-premise deployments are also still applicable to large pharmaceutical companies that already have developed IT infrastructure and have regulatory issues with cloud data storage albeit regressing in terms of relative market share.
By End-User: Customer Segment Analysis
Pharmaceutical and Biotechnology Companies: Largest Segment
Pharmaceutical and biotechnology companies represent the largest end-user segment at USD 1,089 Million in 2025, accounting for 55.0% of total market value, projected to USD 2,357 Million in 2035 with a CAGR of 8.0%. This group includes the giant multinational pharmaceutical companies having large clinical trial portfolios, mid-sized specialty drug companies and the emergent biotechnology companies developing new therapeutics in a wide disease range.
Contract Research Organizations (CROs): Contract Research Operational Effectiveness.
CROs contribute USD 594 Million (30.0%), which involves the CTMS platforms to handle sponsor trials, arrange global networks of sites, monitoring of study milestones, and showing operational effectiveness to their clients. Top CROs such as IQVIA, Syneos Health, PPD and Parexel have extensive CTMS platforms that serve thousands of ongoing studies in areas of therapeutics.
Research and Academic Institutions.
Academic Medical Centers and research institutions are USD 297 Million (15.0%), running investigator-initiated trial, early-stage research and specialized studies with their own operational needs and limited budgets that make cost-effective cloud based-solutions attractive.
By Component: Solution and Service Analysis
Software Solutions: Core Technology Platform
The CTMS software licenses and subscriptions constitute USD 1,188 Million (60.0) of total market value, which comprises the platform licensing and cloud hosting, and software maintenance to support core CTMS functionality such as study planning, site management, patient tracking, document management, financial management and reporting capabilities.
Implementation and Professional Services
Service USD 792 Million (40.0%): Implementation services, consulting, training, and technical support represent the large amount of professional services needed in the successful deployments of CTMS such as requirements gathering, system configuration, data migration, validation, user training, and continuous optimization services.
Regional Market Analysis
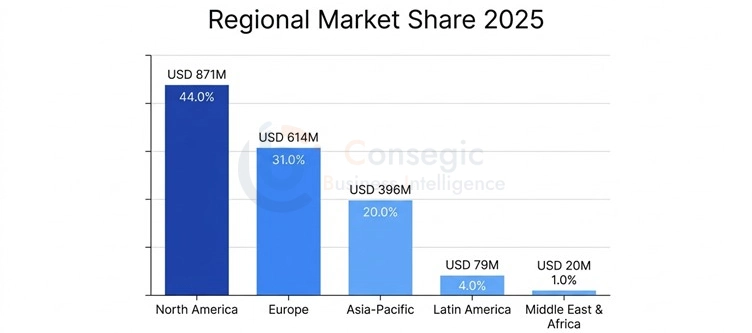
North America: Market Leader and Centrum of Innovation.
The North American CTMS market has USD 871 Million market value in 2025, which depicts 44.0% of the total market value in the world, yet it is projected that the market will be USD 1,885 Million in 2035 with 8.0% CAGR. The United States controls the regional demand with 92.5 percent of the market worth, where there are more or less 48 percent of the global clinical trials, with more than 125,000 active trials and have the highest pharmaceutical market in the world, with USD 595,000 Million of yearly prescription drug sales.
The pharmaceutical and biotechnology firms in North America incur more than USD 98,000 Million annually in R&D and most of the development funds are used on clinical trials. The region has the highest rates of adopting CTMS of 78% among large pharmaceutical companies and 62% among mid-sized biotechnology firms, which are caused by high-levels of trial operations, regulatory compliance needs, and technological leadership.
Europe: Superiority of Regulations and Co-ordination across Countries.
European CTMS market is USD 614 Million in 2025, which is equivalent to 31.0% of the market value globally and it is projected to increase to USD 1,328 Million by 2035 at CAGR of 8.0%. Overall regional demand is made up of 68% by the United Kingdom, Germany, France, and Switzerland, and extensive pharmaceutical industries, good clinical research facilities, and complicated regulatory requirements necessitate sophisticated trial management facilities.
Asia-Pacific: the Rapidly Developing Market.
The CTMS market in Asia-Pacific commands USD 396 Million in 2025, which is equivalent to one-fifth of the global market value, and the growth is very strong, anticipated to reach USD 943 Million in 2035 with the CAGR at 9.1% which is the highest regional growth rate. China is the largest national market with 42.5% regional value, then Japan with 28.5 percent, India with 14.0 percent and other Asian countries and their total value amount to 15.0 percent.
Other Regions
Latin America is USD 79 Million (4.0%), and Middle East & Africa is USD 20 Million (1.0%) with both showing growth potential due to the increasing clinical trial activity, a strong regulatory infrastructure, and cost incentive served as a draw into global pharmaceutical sponsors.
Competitive Landscape and Key Market Players
Market Leadership and Strategic Positioning
Oracle Corporation (United States) - World Market Leader.
Oracle continues to enjoy dominant positions in the market with a projected USD 348 Million CTMS revenues in 2024 as 17.6% market share worldwide on its Oracle Clinical One platform. It offers a full suite of cloud-based unified clinical trial solutions integrating CTMS, EDC, RTSM, safety reporting, and data analytics solutions to more than 850 pharmaceutical, biotechnology, and CRO customers around the world with an estimated 42,000 clinical trials per year.
Veeva Systems Inc. (United States) - Cloud Leader.
As a global market leader, Veeva controls about 14.8% market share as CTMS revenue of USD 292 Million in 2024 with Veeva Vault CTMS being a subset of its overall cloud platform in the life sciences domain. The company has more than 1,200 customers and is highly adopted by biotechnology companies and CROs desiring modern cloud native solutions that can be deployed quickly.
Medidata Solutions (Dassault Systèmes) (United States) - Innovation Leader.
Medidata holds roughly 14.5% of the worldwide market share and the revenue of Medidata Rave clinical trial platform with inbuilt CTMS functionalities are anticipated to reach USD 287 Million in 2024. The company has more than 1,850 customers and 95 percent of the top-25 pharmaceutical companies who are served by the company and manages about 28,000 clinical trials handling 8.5 million patients using its platforms.
Additional Key Market Participants:
- IQVIA Inc. (United States)- 12.5% market share, CRO-integrated solutions
- Parexel International Corporation (United States)- 9.8% market share, service-enabled technology
- Bio-Optronics, Inc. (United States)- 7.2% market share, specialized clinical research focus
- ERT (Clario) (United States)- 6.5% market share, cardiac safety and eClinical integration
- MasterControl Inc. (United States)- 5.8% market share, quality management integration
Recent Industry Developments
Oracle - Artificial Intelligence-Clinical Operations.
In early 2025, Oracle introduced Oracle Clinical One AI, which includes artificial intelligence and machine learning functions of predictive site selection, site recruitment prediction, and risk-based monitoring. The platform uses historical trials data on 42, 000 plus studies to forecast enrollment risk with 89 percent accuracy and the best sites to conduct trials based on prior success record, demographics, and competitive trial environment.
Time Warner- Valeeva Systems - Unified Clinical Platform Enhancement.
Veeva launched Veeva Vault Clinical Suite 25R1 that introduced greater integration between CTMS, eTMF, site payments, and startup applications, cut implementation schedules by 45 percent, and introduced end-to-end clinical data models. The release brings in 150+ customer-requested improvements on the basis of response received by 1,200+ life sciences organizations.
Medidata Solutions - Decentralized Trial.
Medidata declared full extension of its myMedidata patient-facing platform with capabilities of CTMS that allows decentralization of trial management such as scheduling of telemedicine visits, home health coordination, electronic consent, patient-reported outcomes, and direct communication between patient and the study teams.
Clinical Trial Management System Market Report Insights
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2022–2035 |
| Base Year | 2025 |
| Forecast Period | 2026–2035 |
| Market Size in 2025 | USD 1,980 Million |
| Market Size in 2035 | USD 4,285 Million |
| CAGR (2026–2035) | 8.0% |
| By Delivery Mode | Cloud-Based (SaaS), On-Premise, Hybrid |
| By Component | Software, Services (Implementation, Training, Support) |
| By End-User | Pharmaceutical & Biotech Companies, CROs, Academic & Research Institutions, Medical Device Companies |
| By Clinical Trial Phase | Phase I, Phase II, Phase III, Phase IV |
| By Deployment | Enterprise CTMS, Site CTMS |
| By Region | Asia-Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Players | Oracle Corporation, Veeva Systems, Medidata Solutions (Dassault Systèmes), IQVIA, Parexel International, Bio-Optronics, ERT (Clario), MasterControl |
| Report Coverage |
|
Key Questions Answered in the Report
What is the size of the Clinical Trial Management System market? +
The global market of clinical trial management systems is showing a very high potential of growth with an USD 1,980 Million in 2025 and reaching USD 4,285 Million in 2035; the overall growth of clinical trial management systems is expected to be sustained with the growing complexity of clinical trial, regulatory compliance, decentralized trial adoption, pharmaceutical R&D investment growth and digital transformation initiatives of clinical trials in the pharmaceutical, biotechnology and contract research industries.
Which is the most rapidly expanding market in the Clinical Trial Management System market? +
Asia-Pacific is the most rapidly growing regional market with a 9.1% CAGR, enhanced by the rising level of pharmaceutical industries, rising clinical trial activity, governmental policies encouraging clinical research, rising rates of domestic biotechnology companies in China, Japan, India and countries of the southeast.
What are the specific segmentation information that are contained in Clinical Trial Management System report? +
The report also gives detailed segmentation based on Delivery Mode (Cloud-Based, On-Premise, Hybrid), Component (Software, Implementation Services, Training & Support), End-User (Pharmaceutical and Biotech Companies, CROs, Academic and Research Institutions), Clinical trial phase (Phase I, Phase II, Phase III, Phase IV), Deployment (Enterprise CTMS, Site CTMS) and Regional markets with the sub-segmentation and adoption figures.
What are the key players in the market of Clinical Trial Management System? +
Oracle Corporation, Veeva Systems Inc., Medidata Solutions/Dassault Systèmes, IQVIA Inc., Parexel International Corporation, Bio-Optronics, Inc., and a variety of specialized vendors with important platform capabilities and service models in specific therapeutics, customer segments, and geographic markets dominate the competitive landscape (17.6%, 14.8%, 12.5%, 9.8% and 7.2% market share respectively).
Which are the major trends in technology that are influencing the market till 2035? +
Significant technological trends involve the use of artificial intelligence and predictive analytics that can optimally select sites, predicts recruitment, and risk-based monitoring with 85-92 prediction, integrated clinical trial platform to combine CTMS, EDC, eTMF, RTSM, and safety systems that reduce implementation timelines by 35-50, home health coordination, remote patient monitoring, telemedicine, decentralized trial execution, and real-time data visualization and dashboards that give executives insight into the performance of trial portfolios, automated compliance monitoring that reduces regulatory findings