Home > > Pharmaceuticals > > Europe Cell Culture Market Size, Trends, Growth & Forecast 2025-2034
Europe Cell Culture Supplements Market - Size & Industry Share - Growth Trends & Forecasts (2025 - 2034)
ID : CBI_3468 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
Europe Cell Culture Supplements Market Executive Summary
The Europe cell culture supplements sector is a key infrastructure aspect of the biotechnology and biopharmaceutical production platform that facilitate optimal cell growth, maintenance, and therapeutic product expression across various applications. This advanced market includes biopharmaceutical production plants that produce monoclonal antibodies and recombinant proteins, the developers of cell and gene therapies that produce advanced therapeutic medicinal products (ATMPs), academic research centers that conduct cell-based research, and emerging cellular agriculture projects focused on cultivated meat technology.
The market shows strong growth potential, increasing from USD 1.85 billion in 2025 to USD 3.68 billion by 2034 with a compound annual growth rate (CAGR) of 7.9 percent in the 2026-2034 forecast period. This growth is driven by several factors. These include the rapid expansion of cell and gene therapies the explosive growth of cell and gene therapies (with more than 47 advanced therapy medicinal products approved by the European Medicines Agency), the accelerating shift to serum-free and chemically-defined formulations of media used to comply with regulatory requirements, the continued growth of European biologics manufacturing with 285 biological products in clinical development pipelines, and rising investment in cellular agriculture research (more than EUR 420 Million annually out of 35 European companies).
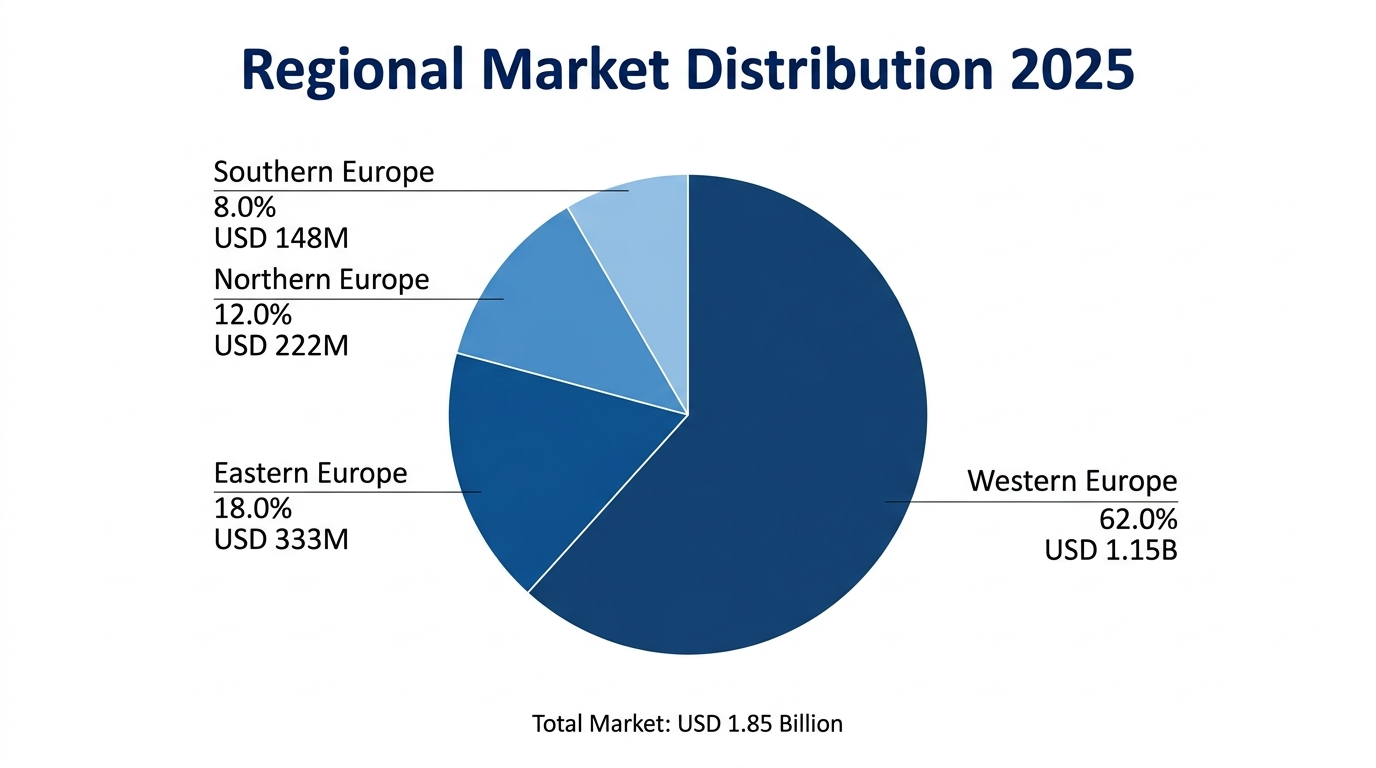
The Western Europe dominates the highest market share of about 62 percent of total market worth which is anchored by the biopharmaceutical excellence in Switzerland where Roche and Novartis conduct their operations, biotechnology infrastructure in Germany with massive manufacturing facilities and cell therapy innovation leadership in the United Kingdom through the golden triangle research corridor. The sub-region with the highest projected CAGR of over 9.5 percent is Eastern Europe, which is driven by growth in contract development and manufacturing organizations, foreign direct investment in bioprocessing plants, and transfer of technology through establishment of cost-competitive operations in the states by multinational pharmaceutical corporations.
Competition is characterized by integrated life science distributors and dedicated biotechnology platforms, with Thermo Fisher Scientific, Merck KGaA, Sartorius AG, Cytiva (Danaher Corporation) and Lonza group as market leaders toward a personalized medicine platform, advances in platform and supply chain resiliency and the provision of animal-component-free formulation development, and media optimization services.
Regional Trends and Insights
Market Growth
- Market Size: USD 1.85B (2025) → USD 3.68B (2034)
- Growth Rate: CAGR 7.9% (2026–2034)
- Indicates strong growth potential
Key Growth Drivers
- Rapid growth of cell & gene therapies
- Shift to serum-free & chemically-defined media
- Expansion of biologics manufacturing
- Rising investment in cellular agriculture
Emerging Region
- Eastern Europe: Fastest growing (CAGR > 9.5%)
- Growth in CDMOs & bioprocessing plants
- Increasing FDI & tech transfer activities
Technological Change and Regulation.
Modern cell culture supplement manufacture uses complex biotechnology platforms such as recombinant protein expression systems using Escherichia coli, Pichia pastoris and Chinese hamster ovary cells with a level of purity of at least 5-15 grams per liter, chromatographic purification systems based on affinity, ion exchange and size exclusion purification techniques with purity levels of greater than 98 percent and advanced quality control systems to ensure compliance with European Medicines Agency requirements and Good Manufacturing Practice requirements.
European biopharmaceutical industry, with an annual revenue of about EUR 285 billion and 820,000 professionals faces the most radical transformation pressures such as the mandatory adherence to European Union requirements on adventitious agent testing, high standards of traceability of raw materials along the supply chain, sustainability demands that have decreased the environmental footprint, replacement of animal-derived ingredients with chemically-defined ones. Opportunities for suppliers of supplements who need to strike a balance between product performance optimization, regulatory compliance documentation, supply chain reliability, and cost leadership when dealing with the growth of biotechnology industry to personalized medicine platforms.
The European investment in the development of cell culture technologies was over EUR 520 Million in 2024, with the largest supplement manufacturers devoting between 6-11 percent of their revenues to research and development efforts in cell therapy and cellular agriculture market applications through recombinant protein alternatives to serum products, optimization of chemically-defined formulations, scalable production, and application-specific supplement portfolios.
Key Drivers:
Cell and Gene Therapy Expansion of Commercialization and Manufacturing.
The market growth velocity is driven by the booming growth and commercialization of the cell and gene therapeutics in Europe, which is an ultimate shift in paradigm between cellular therapeutics which are living cells and the conventional small molecule pharmaceuticals. The European Medicines Agency has suggested approval of 47 advanced therapy medicinal products by 2024 and hundreds of others are in clinical development pipelines in the field of oncology, rare diseases, and in application in regenerative medicine. The cell therapies do not require the secreted proteins in the traditional biologics but instead the modified cells themselves are used as the therapeutic agent, which means that the manufacturing process places a very high-quality standard on the supplements used in cell therapy that are of clinical grade.
An example of this transformation in demand would be CAR-T cell therapy manufacture, with plants run by Novartis producing Kymriah cures and Gilead Sciences producing Yescarta that are used in unison to treat 8,500 patients in European treatment facilities each year. These production processes involve special cytokine cocktails such as interleukin -2 at a concentration of 100-300 international units per milliliter, interleukin -7 and interleukin -15 that promote T-cell growth on patient-samples to therapeutic levels of over 100-600 million cells per therapy. Supplements play a significant role in determining the cost of goods sold on these therapies, cytokines and growth factors constitute 25-35 percent of total cost of production on autologous therapies.
The development of allogeneic cell therapy offers further market opportunities due to the ability to produce standardized productions to allow economies of scale. The companies developing off-the-shelf therapies require reliable and standardized supplement formulations to support the large, and validated cell expansion in bioreactor systems of 50 to 2,000 liters. Scaling from research culture flasks to commercial bioreactors increases supplement consumption by 100–1,000 times per therapeutic batch, exposing scalable, GMP-qualified formulation producers to significant earning chances.
The induced pluripotent stem cell production represents a particular industry within the field of stem cell therapy, where the specified growth factor combinations that preserve pluripotency at the expansion stages and lead differentiation into cell lineages such as cardiomyocytes, neurons, and pancreatic beta cells are necessary to serve specialized supplement demand. European manufacturers of stem cell therapies, such as Cellectis, Celyad Oncology, and Orchard Therapeutics, have manufacturing plants using high-quality supplements such as recombinant albumin, specialized attack factors and small molecule inhibitors with prices that range EUR 500–2,000 a liter of culture media.
Key Restraints:
Complexity of Regulation and Validation requirements for manufacturing changes.
The main key barrier to market entry is the broad based regulatory standards and validation costs involved in introducing the new supplement formulations into the existing biopharmaceutical manufacturing procedures. The European Medicines Agency implements strict change control measures that involve complete documentation of changes, analytical testing, and stability studies in case of changing the component of cell culture media or suppliers of products in the clinical development or commercial manufacturing phase.
The introduction of alternative formulations of supplements into established manufacturing systems requires comparability studies to prove equal cell growth kinetics, productivity levels and product quality characteristics such as glycosylation patterns, charge variants, aggregation profiles as well as biological activity measurements. Such researches are generally 6-12 months of laboratory studies, 50-100 pilot-scale production runs using large amounts of both current and alternative supplements, and detailed analytical characterization using 25-40 different quality attributes measured by using complex analytical techniques such as mass spectrometry, chromatography, and bioassays.
Validating supplement changes would cost between EUR 500,000 and EUR 2.5 million per product in relation to stage of product development, regulatory rule, and complexity of the analytical characterization protocols. These validation requirements impose high switching costs and supplier lock-in effects, and, even considering the risk of supply chain uncertainty, 68 percent of biopharmaceutical manufacturers in this industry have single-source suppliers of vital cell culture supplements despite opportunities to optimize costs.
Qualification of raw material processes require a wide scope of supplier audits, quality agreement negotiations, certificate of analysis documentation, risk assessment and evaluation tasks. Biopharmaceutical producers have approved vendor lists that take 12-18 months to be qualified as a new supplier such as inspection of the facility, analysis of quality system, material testing procedures and verification of regulatory compliance. These barriers present significant barriers to market entry by new suppliers of the supplements, and restrict competition in the already established manufacturing relationships.
Future Opportunities:
Revolution in Cellular Agriculture and Cultivated Meat Production.
There is a large market potential in aiding the nascent cellular agriculture sector that is in the process of producing cultivated meat, dairy, and seafood products based on animal cell culture technologies, which will fundamentally transform the current protein production systems to become sustainable. European cultivated meat industry received EUR 420 Million investment spread across 35 companies developing platforms to produce meat, with the major companies building manufacturing capabilities in Europe to produce cultivated beef, cultivated steak, and cultivated pork products.
These are cultivated bovine, porcine and avian muscle cells in bioreactor systems that give rise to meat products without slaughtering of animals and the use of specialized cell culture supplements that are optimized to food production applications as opposed to pharmaceutical production applications. The production of cultivated meat will require entirely different supplement economies than those associated with biopharmaceutical production, with an 80–95% cost reduction needed to match the prices of conventional meat products (ground meat use) at EUR 8-15 per kilogram.
Pharmaceutical-grade supplements are currently priced between EUR 200 to EUR 800 per liter of culture media whereas cultivated meat manufacture would only cost supplements less than EUR 5-15 per liter to meet target production costs sufficient to be commercially viable. The imperative to this cost compels recombinant protein production on precision fermentation platforms, the alternative growth factors in plants expressed on tobacco and rice, and the formulation development required to satisfy human consumption safety requirements.
Technical opportunities involve that production of plant-derived growth factor alternatives use tobacco or rice or microalgae expression systems that achieve costs of EUR 50-150 per gram as compared to EUR 2,000-8,000 per gram mammalian cell-derived factors, development of recombinant proteins using precision fermentation platforms with scale of over 500,000 liter with costs of between EUR 100-300 per kilogram and hydrolysate-based nutrient formulations that can reduce overall costs.
Alternative proteins are a part of sustainable transformation of the food system and the Farm to Fork Strategy developed by the European Commission promotes alternative protein creation, with Horizon Europe programs providing EUR 185 Million of funding to cellular agriculture research in 2024-2027. The government of the Netherlands had made investments worth EUR 60 Million in cultivated meat infrastructure such as shared pilot production plants and regulatory pathway development that facilitates commercialization schedules. The initiatives, in the form of policies, will provide positive environment to the suppliers of supplements as they design cost-efficient, food-grade formulations which would support commercial scale cultivated meat production estimated at 45,000-65,000 tonnes per year by 2030 across the European markets.
Market Segmentation Analysis
By Product Type: Supplement Category Analysis
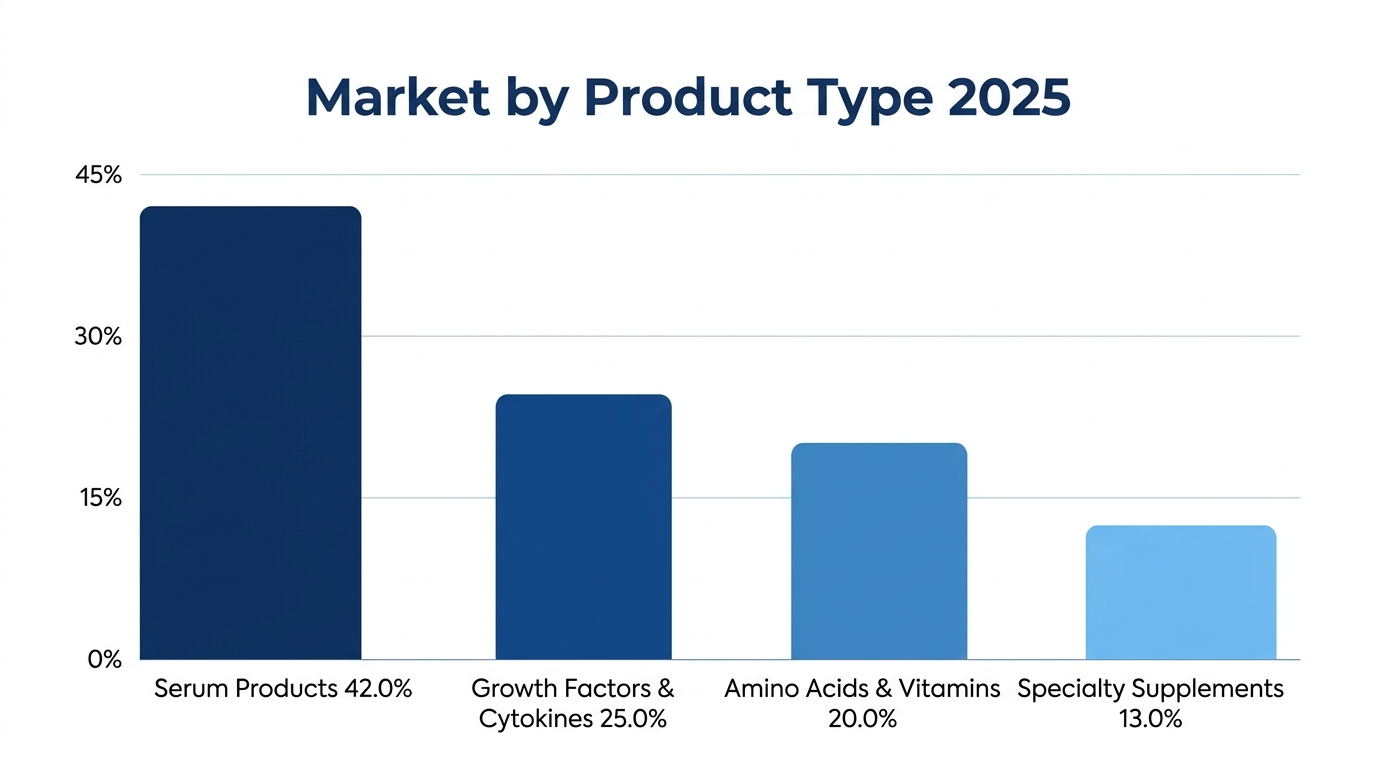
Serum Products: Traditional Market Foundation
Serum products account for USD 777 million (42.0 percent of market value) in 2025, which is expected to grow to USD 1.40 Billion in 2034. Under this category is fetal bovine serum which provides complex mixtures of growth factors, hormones, attaching factors and nutrients that supply various cell types such as insect cell culture systems, horse serum which is used in certain cell lines, such as specialty sera such as human platelet lysate used to manufacture cell therapy in clinical settings.
Despite the ethical concerns related to animal welfare, batch variability problems, and regulatory pressure to get rid of it in clinical manufacturing processes, fetal bovine serum is still the gold standard in research applications. Consumption is 850,000 liters per year in Europe, with the premium serum products being subjected to a lot of testing which includes the 9-point viral testing, mycoplasma testing, endotoxin testing, and hemoglobin testing. The prices vary between EUR 280-650 per liter based on geographic origin, testing comprehensiveness, and performance traits, with the New Zealand and Australian sources being priced high as they have a high disease-free status and accepted by the regulations.
Growth restrictions indicate the continued shift to serum-free options because of regulatory bias towards chemically-defined formulations, batch consistency demands of validated manufacturing process, ethical concerns of animal welfare and security concerns of supply chains following pandemic-driven disruptions of global serum availability and price stability.
Growth Factors and Cytokines: High-Performance Drivers
Growth factors and cytokines USD 462 Million (25.0 percent of market value), which will grow to USD 920 Million by 2034, with fastest growth at 8.1 percent CAGR, due to the growth of cell and gene therapy and optimal production of biopharmaceuticals. This portion contains insulin recombinants that were being substituted by animal-derived insulin at 5-10 milligrams per liter, recombinant transferrin that delivers iron at 5-15 milligrams per liter, epidermal growth factor that facilitates the growth of epithelial cells, fibroblast growth factors that facilitate the growth of mesenchymal cells, and specialized cytokine such as interleukins that are required in the culture of immune cells.
The focus of applications has been biopharmaceutical manufacturing, where regulatory bodies are greatly favoring animal-component-free formulations, cell therapy production that needs clinical-grade supplements that meet GMF requirements, and serum-free media development of Chinese hamster ovary cells, hybridomas, and human cell lines to viable cell densities of 15-25 million cells per milliliter. The replacement of animal products with recombinant ones eradicates batch variability and guarantees enhanced performance and regulatory adherence across manufacturing campaigns.
High-quality cytokines used in the production of CAR-T cell such as interleukin-2, interleukin-7 and interleukin-15 fetch prices of EUR 2,000–8,000 per gram of GMP grade material with a treatment dose of 50-200 milligrams of a combination of cytokines representing supplement cost of EUR 500-3200 per therapeutic dose. Cell therapy manufacturing, which involves volume growth, presents a chance to the suppliers building cost-effective recombinant manufacturing platforms where economies of scale are realized by optimizing fermentation.
Amino Acids and Vitamins: Essential Nutrient Foundation
Amino acid and vitamin supplements constitute USD 370 Million (20.0 percent of the market value), which is bound to increase to USD 662 Million in 2034. This group is characterized by essential amino acids, i.e. leucine, isoleucine, and valine which are needed to synthesize proteins at levels of 0.4- 2.0 millimolar, non-essential amino acids, i.e. glutamine which serves as a source of carbon and nitrogen at levels of 2-8 millimolar, water-soluble vitamins, i.e., B-complex vitamins and vitamin C, which support cellular metabolism, and fat-soluble vitamins, including vitamins A, D, E, and K, which play critical roles in cell signaling, membrane stability, and overall cellular function.
These are used across the entire range of cell culture systems where amino acids are needed during cell growth stages and where protein synthesis requires constant cell supplement in fed-batch protocols. Glutamine is the largest-volume amino acid supplement because of its instability in aqueous solutions necessitating feeding routes or dipeptide substitutes like alanyl-glutamine that presented increased stability and decreased amounts of ammonia. The current production of biomanufacturing plants in Europe involves 450 tonnes of glutamine each year with prices of EUR 45-85/kg on cell culture grade material to pharmaceutical manufacturing requirements.
Specialty Supplements and Attachment Factors: Functional Enhancement
Attachment factors and specialty supplements are USD 240 Million (13.0 percent of market value), and will project USD 478 Million in 2034. This category includes cholesterol to aid membrane production, fatty acids like linoleic acid and oleic acid, phospholipids necessary to maintain membrane integrity, pluronic F-68 to reduce shear stresses in bioreactor systems, fibronectin to support cell adhesion with concentrations of 1-10 micrograms per square centimeter, laminin to assist cell wall attachment to the epithelial cell and collagen to give a three-dimensional scaffold framework to cell growth in the organoid systems.
Commonly used applications are anchorage-dependent cell lines where cell lines need to be cultured in microcarrier-based culture systems that require attachment factors, shear-sensitive cells that can take advantage of pluronic F-68 protection in stir-tank bioreactors operated at 50-150 revolutions per minute, and specialized culture systems such as organoids and tissue engineering applications which need extracellular matrix components that support cell organization and differentiation processes in three-dimensional culture systems.
By Application: End-Use Market Segments
Biopharmaceutical Manufacturing: Dominant Production Platform
The biopharmaceutical production applications are USD 925 Million (50.0 percent of market value), which includes monoclonal antibody manufacturing utilizing Chinese hamster ovary cells in 2,000-25,000 liter bioreactors, recombinant protein manufacturing including therapeutic enzymes and hormones, vaccine manufacturing using viral propagation systems, and biosimilar manufacturing with requiring the same specifications of supplement as the innovator products but optimizing the manufacturing cost with the improvement of process efficiency.
These applications are regulatory compliant with the GMP requirements with a batch-to-batch consistency that facilitates approved manufacturing procedure, supply chain security that involves qualified suppliers that uphold dual sourcing strategy, and total documentation of the regulatory submissions such as chemistry manufacturing and controls component. The production of supplements used in manufacturing processes ranges between 15-35 kilograms every 10,000-liter of bioreactor run with a typical monoclonal antibody manufacturing facility producing 500 kilograms a year which needs EUR 2.5-4.5 million worth of cell culture supplements in upstream processing operations.
Quality standards involve low endotoxin content of less than 0.25 endotoxin units per milliliter, mycoplasma free certification using verifiable test procedures, animal derived component viral safety, and certificate of analysis documentation which will ensure traceability on the supply chain of product originating with source material sourcing to final product release.
Cell and Gene Therapy Manufacturing: Emerging High-Value Sector
Cell and gene therapy applications account for USD 370 million (20.0% of market value) and represent the fastest-growing segment with 11.2 percent CAGR due to commercial therapy approvals and clinical trial expansion in oncology, rare diseases and in regenerative medicine applications. These applications demand clinical-grade supplements with high purity criteria, animal-component-free formula with no risks of xenogeneic contamination and special growth factors to enhance T-cell proliferation, mesenchymal stem cell culture and induced pluripotent stem cell maintenance programs.
Interleukin-2, interleukin-7 and interleukin-15 concentration of 100-300 international units per milliliter are required in CAR-T cell production procedures, and the supplement costs constitute a cost of 25-35 percent of overall production cost of autologous therapies costing EUR 300,000-500,000 per patient treatment. By 2024, European manufacturers of cell therapy were able to produce 8,500 patients and by 2030, it is projected that cell therapy will treat up to 28,000 patients annually as more treatments have regulatory approval and manufacturing capacity is increased by the ability to focus on specific therapies and through production units within the hospital.
The creation of allogeneic therapy opens up opportunities to use standardized formulations of supplements in support of large-scale manufacturing process and companies are developing off-the-shelf products that require consistency and validation of media formulations that can be used to provide economies of scale through centralized production plants that cater to multiple geographic markets.
Academic and Research Applications: Innovation Foundation
Academic and research uses USD 370 Million (20.0 percent of market value), which includes university research laboratories performing basic cellular research, hospitals-based research performance of disease mechanisms, pharmaceutical discovery laboratories screening library of compounds, and contract research organizations offering preclinical testing services. The applications focus on product performance that enables many different cell lines, flexibility to support experimental protocols, technical support to support troubleshooting activities and complete product portfolios to develop methods and optimize the methods.
There are European academic centers that use 12,500 cell culture labs that use supplements to conduct basic research studies on cellular mechanisms, disease modeling studies, drug screening programs, and biomarker discovery programs. The applications of research prove more amenable to the use of expensive specialized supplements when the performance benefits shorten the research schedule, allow new experimental methods, or are characterized by better reproducibility than conventional formulations.
The price sensitivity is highly diverse among academic segments with larger groups of well-established research centers and pharmaceutical alliances advocating high-quality supplements and small labs and student training centers in need of such cost-saving solutions as volume-purchase deals and student discounts provided by large suppliers.
Industrial Biotechnology and Cellular Agriculture: Future Growth Platform
The applications as industrial biotechnology and cellular agriculture are USD 185 Million (10.0 percent of market value) and estimates USD 442 Million USD by 2034 with a CAGR of 10.5 percent. This new market segment involves production of cultivated meat that needs the optimization of supplements at the cost, specific fermentation of alternative protein production, industrial manufacturing of enzymes, bio-fuels development, and dedicated applications such as cellular systems production of cosmetic ingredients.
Applications of cultivated meat require lower costs of supplements than pharmaceutical applications, which can sustain EUR 200-800 per liter, and lead to innovation in low-cost recombinant protein manufacturing, growth factor alternatives derived from plants, and food-grade formulation development which satisfies safety concerns in human consumption applications.
By Cell Type: Culture System Requirements
Mammalian Cell Culture: Dominant Platform Technology
The mammalian cell applications are worth USD 1.26 Billion (68.0 percent of market value), which includes Chinese hamster ovary cell in commercial biopharmaceutical production (65 percent), the human cell lines used in research and in production applications, the monoclonal antibodies generated by hybridomas and the primary cells extracted in tissues used in specialized research applications. These systems need complicated nutrient recipes consisting of amino acids, vitamins, lipids, growth factors, and protecting agents that help sustain eukaryotic cellular metabolism and proteolytic machinery.
The Chinese hamster ovary cells take the center stage in the biopharmaceutical production as it is capable of producing human-like protein glycosylation and strong growth traits within a suspension culture as well as its long-established regulatory history of use in manufacturing. These cells use supplements such as glutamine 2-8 millimolar, glucose 15-25 millimolar and special lipids that aid membrane formation during cell division phases that are rapid to attain viable cell densities of 15-25 million cells per milliliter in minimal and optimized culture systems.
Microbial Cell Culture: Industrial Production Platform
The microbial culture applications are USD 370 Million (20.0 percent of market value), which includes the bacterial systems (Escherichia coli) generating recombinant proteins and therapeutic enzymes, the yeast systems (Pichia pastoris, Saccharomyces cerevisiae) generating the proteins and metabolites, and the filamentous fungi systems used in the production of enzymes and secondary metabolites. These systems have less complex nutrient formulations than mammalian cells but have higher volumes than would sustain fermentation scales of 200,000 liters, which are utilized on an industrial scale.
Systems of bacterial and yeast cultures use supplements with trace elements and sources of nitrogen and carbon substrates in concentrations that maximize the production of particular strains of the organism and a specific product. The applications of industrial fermentation are characterized by cost-effectiveness and scalability, and the supplement formulations are products intended to achieve the greatest productivity at the lowest possible costs of raw materials by maintaining the optimal nutrient ratios and feeding methods.
Stem Cell and Specialty Cultures: Premium Applications
USD 222 Million (12.0 percent of market value) goes to stem cell and specialty culture applications, such as embryonic stem cells, which have been used to do disease modeling and regenerative medicine studies, induced pluripotent stem cells, which have been used to do disease modeling and regenerative medicine research, mesenchymal stem cells, which has been used in cell therapy applications and organoid culture systems, which has been used to model human tissues to drug discovery applications.
Such applications require specific supplements such as defined growth factors that preserve stemness properties, small molecule inhibitors that regulate differentiation programs, extracellular matrix proteins that provide three-dimensional culture models, and attachment factors that permit appropriate cell cell contact and cell differentiation capabilities that are important to sustain physiological cellular functions.
Regional Market Analysis
Western Europe: Market Leadership and Innovation Hub
Dominating Position and Biopharmaceutical Greatness.
In 2025, Western Europe controls USD 1.15 Billion (62.0 percent of market value) and projects to USD 2.28 Billions in 2034, the most substantial regional market is due to the concentration of biopharmaceutical manufacturing facilities, existence of advanced biotechnology research infrastructure, regulatory expertise supporting biologics development, and established relationship with suppliers to guarantee the quality of products and regulatory compliance. Western European market value is represented by integrated pharmaceutical and biotechnology ecosystem in Switzerland, Germany, United Kingdom, and France, which represent 78 percent of the market.
Switzerland holds nearly 28 percent of the Western European market value having large biopharmaceutical manufacturers such as Roche with huge biologics production and Novartis with cell and gene therapy manufacturing business and Lonza Group with 180,000 liters bioreactor capacity at the Visp plant serving global pharmaceutical clients. In 2024, Swiss plants manufactured 85 different varieties of biological products with a total bioreactor capacity of over 450,000 liters in supporting high-quality consumption of supplements, which amount to an average of EUR 385 per liter of bioreactor capacity in the Swiss market, compared to the EUR 245 per liter average in Europe.
Germany has a value of 26 percent on the Western Europe market featuring diversified biopharmaceutical sector; Boehringer ingelheim with 220,000 liters bioreactor capacity contract manufacturing services, Merck KGaA with biosimilars and life science reagent production and Sartorius with bioprocessing equipment and consumables. In 2024, the German facilities manufactured 95 biological products with a powerful presence in the biosimilars that captures 32 per cent European biosimilar market value. The nation has 145 biotechnology firms which is engaged in the cell culture research, 28 contract development and manufacturing organizations which is offering its services to pharmaceutical clients who need special formulations of supplements.
United Kingdom is a 15 percent share of Western European market value, with an excellent academic research base (85 universities) performing cell biology research, pharmaceutical firms such as GSK and AstraZeneca developing biological therapeutics, and cell therapy firms such as Autolus Therapeutics and Adaptimmune producing CAR-T products. The Golden Triangle of London, Oxford, and Cambridge generate a focused demand of performance supplements to the highest performance in relation to the cutting-edge research and clinical development programs. The UK has strong demand in the supplement sector with research excellence and specialization in manufactories despite the regulatory complications associated with Brexit.
The pharmaceutical companies such as Sanofi coming up with biological products, academic research centers undertaking research on stem cell and government funding on research commercialization and manufacturing development make up 9 percent of the Western European value (France).
Eastern Europe: Rapid Growth Prospectus.
New manufacturing and contract services centre.
Eastern Europe has USD 333 Million (18.0 percent of market value), which is growing to USD 795 Million by 2034, with the highest regional growth rate of 9.5 percent CAGR due to the growth in contract manufacturing, biosimilar production and the rise in research capacity in the region through increased expenditure by the pharmaceutical companies on facilities in the region that has cost advantages and growth in capacity to research by the academic institutions. The core biotechnology cluster that is enticing foreign investment is Poland, Czech Republic, Hungary and Romania due to the availability of skilled workforce, low labor costs averaging EUR 8-12 per hour as opposed to EUR 35-45 in Western Europe and the free market in Europe which has removed trade barriers.
Eastern European markets are led by Poland who has 32 percent share of the region value with the presence of biopharmaceutical manufacturing facilities run by Polpharma Biologics which manufactures biosimilars and contract manufacturing service with 50,000 liters bioreactor capacity, Mabion developing and manufacturing monoclonal antibodies to serve the European markets and international contract manufacturing organizations such as Samsung biologics also establishing operations in Europe. The Polish plants show a growth rate in the consumption of supplement at 15.5 percent per annum due to the capacity growth and transfer of technology by the parent companies in the Western Europe that are carrying out the proven manufacturing processes.
Czech Republic has 24 percent of the Eastern European market value, and its contract research organizations such as Sotio are carrying out cell therapy research and development programs, academic institutions such as Charles University have cell biology laboratories with modern equipments and pharmaceutical companies have established research centers that are exploiting skilled scientific workforce and low cost of operation. The nation has competitive prices of supplements at an average of 15-25 percent lower than that of the Western Europe, and with quality standards, having passed the ISO certification and met the requirements of the European Union.
Hungary is 18 percent of Eastern European value with pharmaceutical manufacturing business of Richter Gedeon creating biological products, academic research at Semmelweis University and University of Szeged conducting cellular research programs, government investment in biotechnology infrastructure by EUR 145 Million national biotechnology strategy facilitating research commercialization, manufacturing development, and technology transfer programs in multinational pharmaceutical corporations.
The Romania is contributing 12 percent of the regional value by having emerging biotechnology sector comprising of academic research centres, pharmaceutical firms establishing their manufacturing set up, as well as government support on the development of life science through European Union structural funds, and national innovation programmes that encourages technology transfer and building industry.
Southern Europe: Research Excellence and Specialist Applications.
Southern Europe has USD 148 Million (8.0 percent of market value), which is expected to project USD 294 Million in 2034. Italy is the top South European value with 42 percent of the value comes through pharmaceutical firms such as Kedrion Biopharma which is engaged in manufacturing plasma-derived therapeutics, Takis Biotech which is a company engaged in developing DNA vaccinations and immunotherapy products, and academic excellence through University of Bologna, Sapienza University of Rome and University of Milan in their cell biology research and biotechnology development initiative.
Spain is the largest contributor of Southern European value with research institutions such as Barcelona Biomedical Research Park that hosts several biotechnology firms, pharmaceutical activities such as Grifols that manufactures plasma product as well as emerging cell therapy firms generating innovative treatments. The favorable environment in terms of supplement demand growth is provided by the government assistance in terms of the national research programs and the European Union funding.
Northern Europe: Best Research Markets.
Northern Europe USD 222 Million (12.0 percent of market value), growing to USD 442 Million in 2034. The area has had the highest amounts of per-capita investment in research with Sweden coming out first through Karolinska Institute, a medical research center, Uppsala University a place that runs biotechnology courses, and pharmaceutical firms such as Swedish Orphan Biovitrum that develops rare diseases treatment.
Denmark provides by Novo Nordisk biologics production which manufactures diabetic treatments, University of Copenhagen research programs, and biotechnology firms such as Bavarian Nordic which manufacture vaccines. These markets focus on high-end supplement products that can assist in the state-of-the-art research use and specialized manufacturing processes and are ready to take premium prices in order to gain higher performance and technical support services.
Market Competitive Environment and Major Marketplace Competitors.
Strategic Positioning and Leadership in the Market.
Thermo Fisher Scientific (United States - Europe Operations) - Life Science Leader.
Thermo Fisher Scientific continues to dominate the cell culture supplement market in Europe with projected European revenues of EUR 315-355 Million, which is approximately 24-27 percent market share in supplements. The company functions under integrated Gibco and HyClone brands with complete product portfolio such as classical sera, recombinant proteins, amino acids, vitamins, cytokines, and specialty supplements that are used in a variety of applications ranging between basic research and commercial biomanufacturing activity.
Merck KGaA (Germany) - One-Stop-Shop Solutions.
Merck KGaA European cell culture supplement activities would attain a projected EUR 275-315 Million revenues in the year 2024 with an estimated market share of about 20-23 percent with a complete range of products and integration of bio-processing solutions. The company conducts its business through Life Science division that sells supplements under Sigma-Aldrich, Millipore, and BioReliance brand with core competence in recombinant proteins, chemically-defined media components and custom formulations used in pharmaceutical manufacturing and research.
Sartorius AG (Germany) - Integration Specialist Bioprocessing.
Sartorius AG cell culture supplement activities enable the generation of estimated EUR 205-245 Million European revenues in 2024 with a specific market share of approximately 14-17 percent, that is in the sphere of integrated bioprocessing solutions, where supplements are combined with bioreactor systems, single-use technologies, and process analytical instruments. In 2021, the company purchased Biological Industries that added to the portfolio of supplements and acquired proprietary serum products and specialty formulations to aid a wide variety of cell culture applications.
Recent Industry Developments
Animal-Component-Free Formulation Innovation (2024-2025).
Large supplement producers boosted the development of animal-free products in major areas of regulatory preference, security concerns of the supply chain, and ethical issues. Thermo Fisher Scientific has introduced OptiPro serum-free medium, which is optimally designed to culture Chinese hamster ovary cells to viable cell densities of 18-24 million cells per milliliter and antibody titers of 6-9 grams per liter, and has done so without the use of fetal bovine serum, by using optimal combinations of recombinant proteins and growth factors.
Merck KGaA launched Cellvento 4CHO-X, chemically-defined medium formulation, which contained recombinant insulin, transferrin, and albumin as substitutes to all animal derivatives and fulfilled the GMP standards of commercial use in manufacturing. The formulation has proved effective technology transfer over 15 biopharmaceutical manufacturing plants over the year 2024 assisting in the production of 8 different monoclonal antibody products that have the regulatory agency approval of the media change under comparability protocols demonstrating the identical characteristics of product quality.
Sartorius has further developed Xell platform consisting of animal-free supplements to stimulate mesenchymal cells proliferation to be used in cell therapies with 45-fold growth over 14 days and preserving differentiation capabilities and surface marker expression patterns vital to therapeutic activity. It was clinically-grade certified and could be used in GMP production of autologous and allogeneic cell therapies with detailed documentation to support regulatory submissions.
Europe Cell Culture Supplements Market Report Insights
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2022–2034 |
| Base Year | 2025 |
| Forecast Period | 2026–2034 |
| Market Size in 2024 | USD 1.71 Billion |
| Market Size in 2025 | USD 1.85 Billion |
| Market Size in 2034 | USD 3.68 Billion |
| CAGR (2026–2034) | 7.9% |
| By Product Type | Serum Products (42.0%), Growth Factors & Cytokines (25.0%), Amino Acids & Vitamins (20.0%), Specialty Supplements (13.0%) |
| By Application | Biopharmaceutical Manufacturing (50.0%), Cell & Gene Therapy (20.0%), Academic & Research (20.0%), Industrial Biotechnology (10.0%) |
| By Cell Type | Mammalian Cells (68.0%), Microbial Cells (20.0%), Stem Cells & Specialty (12.0%) |
| By Region | Western Europe (62.0%), Eastern Europe (18.0%), Northern Europe (12.0%), Southern Europe (8.0%) |
| Key Players | Thermo Fisher Scientific, Merck KGaA, Sartorius AG, Cytiva, Lonza Group, Corning Life Sciences, Biological Industries |
| Report Coverage |
|
Key Questions Answered in the Report
What is the size of the market of the cell culture supplements of Europe and what is the growth driver of the market? +
The European cell culture supplements market portrays strong growth potential with a 2025 value of USD 1.85 Billion and a growth projection of USD 3.68 Billion in 2034 with a growth rate of 7.9 percent CAGR. This expansion represents a long-term demand of commercialization through cell and gene therapy production of 47 advanced therapy medicinal products and the rise in biopharmaceutical manufacturing with 285 biological products such as monoclonal antibodies and recombinant proteins, shift toward animal-free formulations in compliance with regulatory requirements and supply chain security, and novel cellular agriculture uses necessitating cost-effective supplements. It has a market of about 12,500 research laboratories, 285 commercial biomanufacturing plants and 47 cell therapy manufacturing plants that utilize supplements in various applications
There are a few key sources of market growth, what are these? +
Commercialization of cell and gene therapy has become the most influential force in generating a specialized need of clinical grade supplements that help in the production of CAR-T cells, cell expansion, and growth of stem cells, and production of gene modified cells. The European plants serve 8,500 patients per year using cell therapies that involve cytokines and growth factors that amount to 25-35 percent of the manufacturing expenses per patient therapy. The growth of biopharmaceutical manufacturing helps in long-term supplement consumption where the European plants have a total of 1.8 billion liters of bioreactor capacity that consume supplements at 15-35 kilograms per 10,000-liter generated production. Animal-component elimination regulatory pressure is leading to shift to recombinant protein supplements with a 8.1 percent CAGR and new market opportunities with lower costs of supplements below EUR 5-15 per liter are being created by new cellular agriculture applications that can withstand EUR 200-800 per liter pharmaceutical applications.
What are the best performing markets in the region? +
Western Europe holds the largest market share of 62.0 percent with USD 1.15 Billion value characterized by the concentration of biopharmaceutical manufacturing with the presence of 3 manufacturers operating in Switzerland with Roche, Novartis, and Lonza facilities accommodating 85 biological products, Germany is a key contributor, with world-leading cell therapy innovation centers in the form of the Golden Triangle research corridor. Eastern Europe has the highest growth rate at 9.5 percent CAGR with USD 333 Million market value due to contract manufacturing, biosimilar growth, and production, pharmaceutical company, investment into regional manufacturing operations whereby labor costs is lower in the East at an average of EUR 8-12 per hour with labor costs in Western Europe at EUR 35-45.
What are the competitive forces that define the market structure? +
The market structure of the European cell culture supplements has moderately concentrated competition where the three leading suppliers control 65-70 percent market share. Thermo Fisher Scientific is the market leader with a 24-27 percent share on combined Gibco and HyClone brands with complete product lines and worldwide supply chain source of 78 percent of European biopharmaceutical manufacturing houses. Merck KGaA has 20-23 percent share in local European production and regulatory skill in supporting 72 percent of biosimilar production activities. Sartorius owns 14-17 percent stake with a focus on integrated bioprocessing solutions comprising of supplements and bioreactor systems and single-use technologies. Competition is stiffer in animal-component-free formulations, tailored media development services, and strategic liaisons with biopharmaceutical companies and cell therapy developers that need to have specific formulations and technical aid.
What will the future market development be like via technology trends? +
Other major technological advances have been the development of animal-component-free formulations based on recombinant proteins with growth rates of 8.1 percent CAGR, which avoids fetal bovine serum without impairing cell culture performance through the optimization of recombinant insulin, transferrin, and albumin combination to meet GMP requirements to manufacture in significant commercial quantities. The development of chemically-defined media allows reproduction of manufacturing processes with known component concentrations to enable compliance with regulatory requirements and validation of the process. Recombinant growth factors made on precision fermentation platforms cost between EUR 100-300 per gram to sustain cellular agriculture economics needed 80-95 percent cost savings over pharmaceutical uses. Optimization services on custom media with design of experiments methodologies and accelerated high-throughput screening shortens the entire formulation development processes as well as enhances cell growth behavior and productivity improvement up to a 4-6 months period.
