Home > > Medical Devices > > Europe Sequential Compression System Market Size, Trends | Forecast 2034
Europe Sequential Compression System Market - Size, Share, Industry Trends, and Forecasts (2025-2034)
ID : CBI_3487 | Updated on : | Author : Yogesh K | Category : Medical Devices
Market Scope & Overview
The market of the Europe sequential compression system is an industry that is specialized in the medical devices and vascular therapy industry and includes intermittent pneumatic compression devices that prevent deep vein thrombosis, control lymphedema and improves venous circulation by sequentially inflating and deflating air chambers of therapeutic garments. These systems play important functions in hospital facilities, rehabilitation facilities, ambulatory surgery facilities, and increasingly in home-based healthcare facilities, with important functions in both acute prophylaxis purposes and chronic circulatory management purposes.
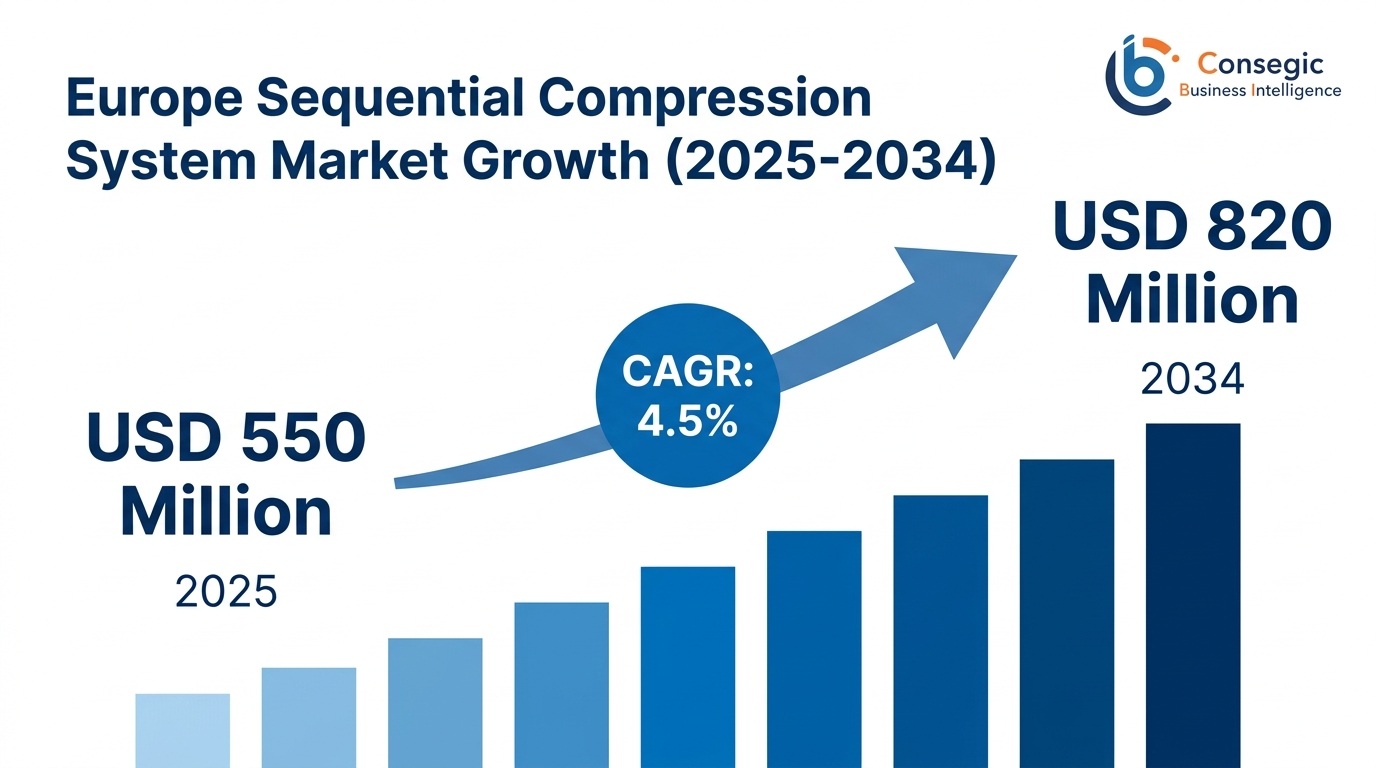
The market has proven great clinical uptake with a valuation with USD 520 million in 2024 and expected to reach USD 550 million in 2025, which is the base year in this comprehensive analysis. These market expectations represent a strong growth to USD 820 million in the year 2034 and the compound annual growth rate is 4.5 percent in the 2026-2034 forecast period. This trend is rooted in the aging demographic profile of Europe together with the more than 20 percent aged 65 years and above population of the European population, rising numbers of surgical procedures of more than 32 million operations every year throughout the continent, the strict clinical guideline to use mechanical prophylaxis to prevent venous thromboembolism, and the rising trend toward home-based structures of healthcare delivery.
Key Regional Insights
- Western Europe: USD 302.5 Million (2025) → USD 451 Million (2034); 55.0% share
- Germany: ~34% share; driven by advanced hospital infrastructure, strong reimbursement system, and high adoption of compression systems
- France: 23% share; growth driven by universal healthcare, surgical safety programs, and rising outpatient care
- Eastern Europe: USD 132M (2025) → USD 213.2M (2034); fastest CAGR of 5.8%
- Poland: 32% share in Eastern Europe; driven by healthcare reforms and hospital upgrades
- Romania & Czech Republic: 28% share; growth driven by infrastructure investments and expanding home healthcare adoption
Technological Evolution and Clinical Integration
Modern sequential compression devices have incorporated the latest engineering designs such as microprocessor-controlled pressure regulation that provides compression levels of 30-120 mmHg across several independent chambers, programmable inflation programs with adjustable time intervals, wireless connection thus allowing remote patient monitoring via safe cloud network and portability of battery powered devices that enable ambulatory treatment regimes. Hospital grade systems have multi-patient capability with 4-8 concurrent treatment channels, automated pressure calibration within a +-2mmHg range, and connectivity with electronic health record systems to provide all clinical records.
The European healthcare system that serves a population of seven hundred million people with an estimated 1.8 million hospital beds and a healthcare expenditure of over EUR 1.9 trillion is faced with the growing challenges such as an increase in the incidence of venous thromboembolism estimated to affect 544,000 patients annually in the EU member states, healthcare-associated complications that have incurred billions of extra costs due to increased hospital stay, and workforce pressures due to an estimated 230,000 positions that become vacant by 2030. These systemic pressures present both operational challenges and market opportunities to manufacturers of sequential compression systems as they develop solutions that meet clinical efficacy requirements while supporting healthcare efficiency and cost optimization.
Executive Summary
The market of the Europe sequential compression system is a mature and dynamically changing segment that is fueled by the evidence-based clinical protocols and demographic demands. The existing market value of USD 550 million in 2035 is based on developed adoption trends in institutional healthcare settings, and the future value of USD 820 million in 2034 is expected due to the increased use in home healthcare, development of technology to portable connected devices, and a deeper understanding of the value of compression therapy in the management of chronic diseases.
The solid growth of the market is supported by the high clinical evidence in the effectiveness of the mechanical prophylaxis, and studies showed that the development of post-surgical DVT can be reduced by 40-60 percent when the compression systems are used based on the recommended protocols. Sequential compression systems are officially integrated in the European and national clinical guidelines of moderate to high-risk surgical patients during prophylaxis, generating systematic demand in healthcare facilities. Venous thromboembolism is estimated to cost economies of EU member states up to EUR 1.5-13.2 billion a year, which has given strong cost-effectiveness grounds as to why prevention compression therapy should be adopted.
At the same time, the market has limitations such as budget constraint in the health systems that are mainly financed by governments, variation in reimbursement between the European nations especially when it comes to home-based application, and pricing pressure on competing products being cheaper. All these influences the growth projections but promote innovation towards value-based solutions that show clear clinical and economic results.
Key Market Driver
Aging Population Demographics and Rising Surgical Volumes with Mandatory VTE Prevention Protocols
The leading market factor revolves around the fast-ageing population make up in Europe and subsequent growth in surgical operations that entail systematic deep vein thrombosis prevention. According to the statistical data of Eurostat, in 2023, the residents of 65 years and older age group comprised 20.8 percent of the EU population, and this is projected to be at 29.4 percent in the year 2050, and it is estimated to have 149 million aged citizens. This population shift is directly proportionate to exponentially higher thromboembolism risk because the frequency of venous thrombosis is 2-3 per 1,000 persons/year in the general population whereas in patients over 80 years of age, it reaches 20 per 1,000.
The total volume of surgical procedures in the European hospitals surpassed 32.4 million surgery procedures in 2023 of which orthopedic surgeries were 4.8 million surgeries, cardiovascular surgeries were 2.1 million surgeries and oncological surgeries were 3.6 million surgeries. Post-surgery DVT is a life-threatening complication, and the untreated patients have 40-60 percent chances of developing thrombosis after having undergone major orthopedic surgeries like hip and knee replacement. Medical practices provided by the European society of cardiology, national health authorities and professional surgical societies require moderate to high-risk surgical patients to receive mechanical prophylaxis, establishing a homogenous institutional demand in healthcare institutions.
The costs associated with the economic effect of the venous thromboembolism complications are high to the European health care systems with direct medical cost in EU member countries estimated to be EUR 1.5-13.2 billion per year. Hospital-acquired DVT prolongs the length of stay by 5-7 days, incurring an extra cost of EUR 3,000-8,000 per episode and eating up scarce bed space. It is these financial factors that motivate institutional adoption of preventive technologies and sequential compression systems have shown good cost-effectiveness ratios of EUR 8,000-15,000 per quality-adjusted life year saved, which are within the reasonable ranges of health technology assessment agencies in European nations.
Demand in the market is further enhanced by regulatory frameworks that aid systematic DVT prevention. The European Commission Patient Safety and Quality of Care program set standards on reducing hospital-acquired complications and the member states adopted national action plans, including mechanical prophylaxis guidelines. France and other countries such as Germany provide EUR 45 million specifically to support surgical safety improvement initiatives in 2023, and Germany requires hospitals to report their quality, which creates compliance-related demand on compression systems with built-in monitoring and documentation systems.
Key Market Restraint
Healthcare Budget Constraints and Fragmented Reimbursement Frameworks
The main market constraint will be the high capital requirements required to carry out full implementation of the sequential compression system as well as the constant budgetary constraint that faces European healthcare institutions. Multi-patient compressor units of professional quality cost between EUR 8,000-25,000 each and complete ward-wide installations cost 15-30 compressor units per ward, or capital investments of EUR 120,000-750,000 per facility. These costs are competing on limited funds on capital equipment often amounting to 3-5 percent of total hospital operating budgets, and it requires hard choices of prioritization among many competing medical technologies investments.
The European healthcare systems are working with a tight fiscal sector, where the rate of growth in the public health expenditure of up to 1.8-2.4 percent per year is unable to keep up with the inflation of the healthcare systems and rising demand of the services. Since the recent economic pressures, countries such as Italy, Spain and Portugal have adopted austerity policies that have cut the budgets of the healthcare capital investments by 15-25 percent, which has resulted in the creation of deferred equipment replacement cycles and lengthy procurement timelines. Hospital administrators have difficult resource allocation choices, which usually involve allocating more resources to diagnostic imaging technologies or surgical robotics or critical care technologies that are believed to bear more clinical impact or revenue potential than prophylactic compression systems.
The sequential compression therapy is cost-based on the recurring cost, and the cost comes with other financial burdens other than the initial capital expenditure. Single use compression dressings, which are a part of the infection control measures and need to be fitted to the patient, are priced between EUR 15-45 each set, and a patient with an average course of treatment in a hospital facility will need between 3-8 sets of this compression garment. Garments alone are costlier, EUR 270 540,000 per year, which is based on a 500-bed hospital with 12,000 surgical operations annually. Compression unit maintenance agreements are priced at EUR 800-1500 a year per unit on average and employee training programs and quality control measures demand extra resource over other clinical demands.
Financial barriers are further hindered by the reimbursement limits especially in home healthcare whereby sequential compression therapy on chronic diseases is subject to unequal covers across European nations. Whereas hospital-based DVT prophylaxis is reimbursable as a diagnosis related group (DRG) payment, standalone home compression therapy faces inconsistent coverage models. In other countries such as Germany a standard of statutory health insurance covers prescribed indications on a comprehensive basis, but in other countries the requirements are a 10-30 percent co-payment or a particular population including chronic disease treatment procedures, limiting access to the market in terms of a home-based application or chronic disease treatment protocol.
Key Opportunity
Home Healthcare Transition and Digital Health Integration
The most significant growth potential occurs due to the rapid shift to the models of home-based healthcare delivery and the combination of the sequential compression systems with remote patient monitoring systems. The optimization of care settings is becoming a prior concern of the European healthcare systems as home healthcare spending is increasing at the yearly rate of 6.8 percent, reaching the overall total of EUR 68 billion in the continent in 2023. This strategic change is in response to the capacity limits of hospitals, patients desiring home-based care where 73 percent of the Europeans preferred home care when clinically appropriate, and cost benefits of home healthcare is proven where 30-50 percent of savings were made in comparison to institutional care in terms of appropriate medical conditions.
Sequential compression therapy shows a specific ability to be used in home healthcare transition, as a treatment of chronic conditions such as lymphedema which has been seen to affect about 3.5 million Europeans, chronic venous insufficiency that affects 25-40 percent of adult populations, and post-surgical care that is increasingly provided in outpatient care. The technological innovations make this possible as they include portable compression devices that are easy to carry (2.5-3.8 kilograms), batteries that can be charged to last up to 8-12 hours of continuous use, and user-friendly interfaces that permit patients to administer themselves with minimal training. The market analysis shows that the sales of home healthcare compression systems have an 8.4 percent CAGR, which is much higher in comparison with the growth rates of the institutional segments.
Integration Remote patient monitoring offers strong value propositions to healthcare providers, payers, and patients by providing better clinical monitoring and increased treatment adherence. Modern compression systems are provided with cellular connection that sends real-time treatment adherence, pressure delivery verification and usage trends to the clinical monitoring centres. The capabilities can be used to handle the basic adherence difficulties and research has shown a low compliance rates of 45-60 percent to prescribed home compression therapy with non-connected devices that are used traditionally. Linked systems show improvement rates of adherence to 75-85 percent with automated prompts to patients, alerts to clinicians on non-compliance trends, and interactive patient engagement capabilities which both facilitate lasting therapy compliance.
The healthcare systems achieve significant values such as lower readmission rates (18-25 percent of patients under surveillance), fewer visits to the clinic leading to the allocation of resources, and early diagnosis opportunities avoiding the development of complications that may need costly emergency treatments. There is regulatory change that helps in this opportunity path, and new EU Medical Device Regulation (2017/745) introduces detailed systems of software as medical devices applications and connected health technologies. Healthcare systems such as NHS England and Haute Autorite de Sante in France established formalized reimbursement systems of remote patient monitoring based on payment models with ranges between 15-35 EUR/patient/month of qualified monitoring services, which is a good economic incentive to implement connected compression system.
Market Segmentation Analysis
By Product Type: Technology Platform Analysis
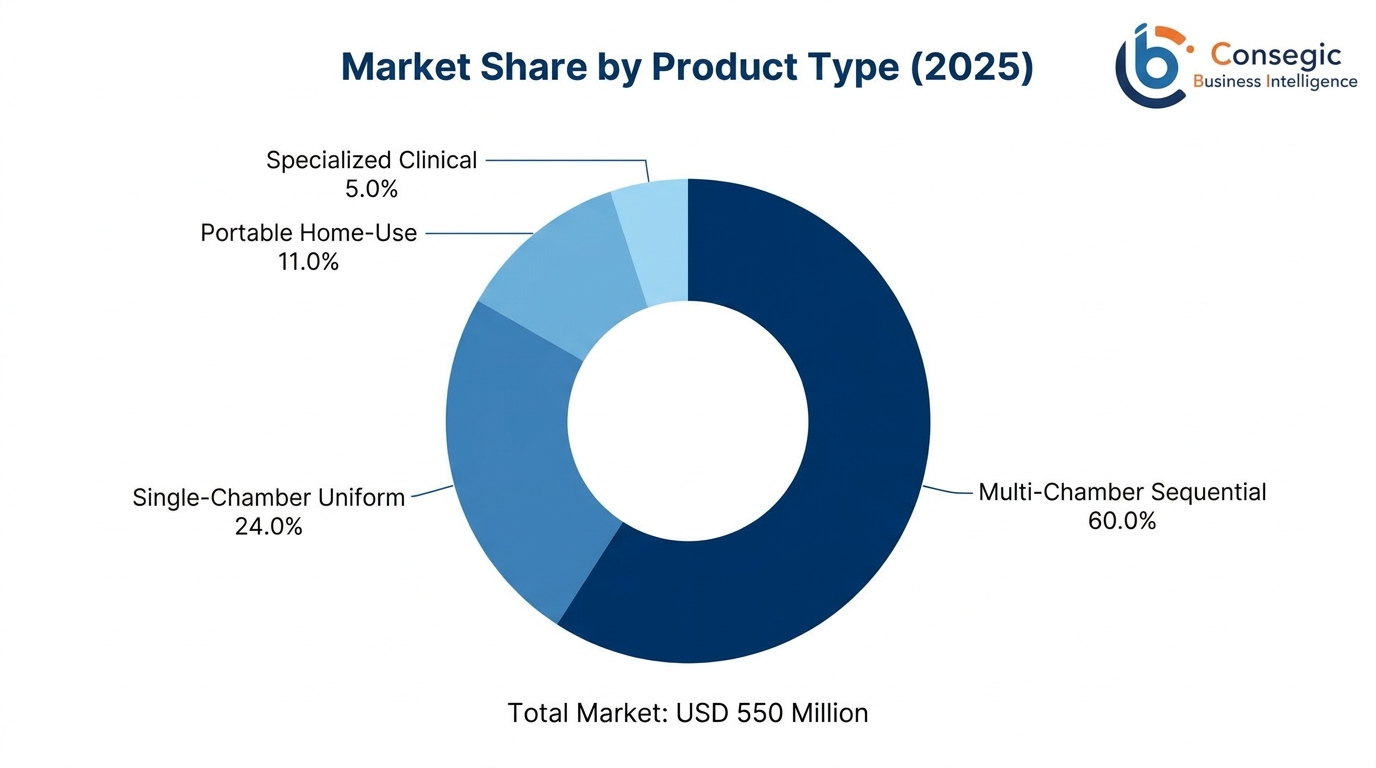
Multi-Chamber Sequential Systems: USD 330 million market leadership position with a 60.0 percent market worth in 2025 at a rate of USD 492 million in 2034. These advanced systems have 4-12 free air chambers, which are positioned in a sequential manner along limbs, which provide a graduated pattern of compression which practically replicates the physiological action of the pump of the heart muscle. Its uses include extensive hospital DVT prophylaxis initiatives, post-operative reconvalescence therapy, and state-of-the-art lymphedema treatments, and the pressure of the compression can be programmed at 30-80 mmHg, and the inflation patterns can be tailored entirely. The preference of clinics is based on proven high limits of efficacy in enhancing venous flow, where controlled tests have shown that 180-240 percent increases in peak venous velocity have been achieved over single-chamber options, which indicates evidence-based implementation in the high-acuity clinical environments.
Single-Chamber Uniform Systems: USD 132 million which represents 24.0 percent of market value, which provides cost-effective applications and minimum prophylaxis among smaller medical centers and budget-restricted institutions. These systems provide consistent compression throughout garments, which are appropriate in the context of low-risk patient groups and centers focused on the basic compliance with mechanical prophylaxis. The 40-60 percent pricing benefits over that of 40-60 percent compared with other multi-chamber units encourage their use in the community hospitals, extended care units and resource-restricted healthcare setting without compromising the much-needed clinical effectiveness to the right patient group.
Portable Home-Use Systems: USD 60.5 million representing 11.0 percent of market value exhibits best growth trend with a CAGR of 7.1 percent over the course of the forecast. This booming market is being used in the home healthcare markets with small sizes, easy user controls and long battery life. Growth in the market due to the chronic disease management needs, especially lymphedema that affects 1 in 1,000 people in Europe and post-surgery home recovery measures endorsed by the enhanced recovery after surgery programs that promote early hospital discharge with subsequent mechanical prophylaxis at home.
Specialized Clinical Systems: USD 27.5 million of 5.0 percent of market value, includes advanced units for use in intensive care, extended-size bariatric patient systems, pediatric-specific devices with the right pressure adjustments, and systems (research grade) that support clinical investigation protocols. These are specialized systems that will attract high pricing between EUR 15,000-35 000 per unit that will serve the niche clinical needs in the tertiary care centres, academic hospitals and specialized treatment centres with specific needs that need more capabilities than typical application of compression therapy.
By Application: Clinical Use Case Analysis
Deep Vein Thrombosis Prevention: Leading application segment of USD 247.5 million of 45.0 percent of market value, used in systematic post-surgical prophylaxis programs, immobilized patient management programs, and high-risk medical patient groups across acute care environments. The sustained demand in this segment is due to annual DVT incidence of about 544,000 cases in Europe and the requirement of mechanical prophylaxis by clinical guidelines. The penetration in the surgical departments of hospitals goes up to 65-80 percent adoption of hospital compression therapy with the compression therapy being the established standard of care in moderate to high-risk groups of patients on the basis of evidence-based clinical guidelines.
Lymphedema Management: USD 192.5 million representing 35.0 percent of market worth, which serves chronic lymphatic system dysfunction in an estimated 3.5 million Europeans with either primary or secondary lymphedema conditions. This application is showing strong growth of 6.2 percent CAGR because of the rising clinical awareness, advanced diagnostic ability and the growing home health uptake that is enabled by the rising reimbursement trends. The norm regimens include long durations of compression therapy, 1-2 hours a day, which are generally extended treatment interventions, placing the load on the home-use systems to provide better patient comfort and compliance.
Chronic Venous Insufficiency Treatment: USD 110 million of 20.0 percent market value, USD 110 million of 20.0 percent of the market value, serving the patients with the venous valve dysfunction, post- thrombotic syndrome, and need of managing the venous ulcerators. The prevalence of the condition ranges between 25-40 percent of adult populations in Europe and the prevalence rate has been found to rise considerably with age. Sequential compression therapy offers conservative management alternatives with proven venous ulcer healing rates of 60-75 percent among the standardized 12-week treatment regimens that justify clinical implementation in vascular care delivery and wound care facilities in the European health care systems.
By End-User Segmentation
Hospitals and Surgical Centers: Main end-user group that needs high-capacity multi-surgical systems, standardized clinical practice, a multi-faceted staffing training, and integrated quality assurance frameworks to support systematic prophylaxis implementation mechanisms in various patient groups and surgical specialties.
Rehabilitation and Long-Term Care Facilities: The Rehabilitation and Long-term care market is a growing segment that is following the compression therapy in providing long-term care to the chronically-ill patients with the focus on ensuring the durability of the system, ease of use by the diverse skill levels, and affordability of the solution to ensure the provision of long-term care to the patients in the resource-conscious care settings.
Home Healthcare Patients: The most rapidly expanding category that requires portable systems with less complex operation, more comfort features to the patient, remote monitoring tools, and interactive patient education tools that would launch effective self-administered therapy protocols in their homes.
Ambulatory Surgical Centers: new area dedicated to perioperative prophylaxis on the outpatient, portable systems must be done to serve pre-operative, intraoperative and immediate post-operative usage to support enhanced recovery regimens and same-day discharge programs.
Regional Market Analysis
Western Europe: Market Leadership and Premium Positioning
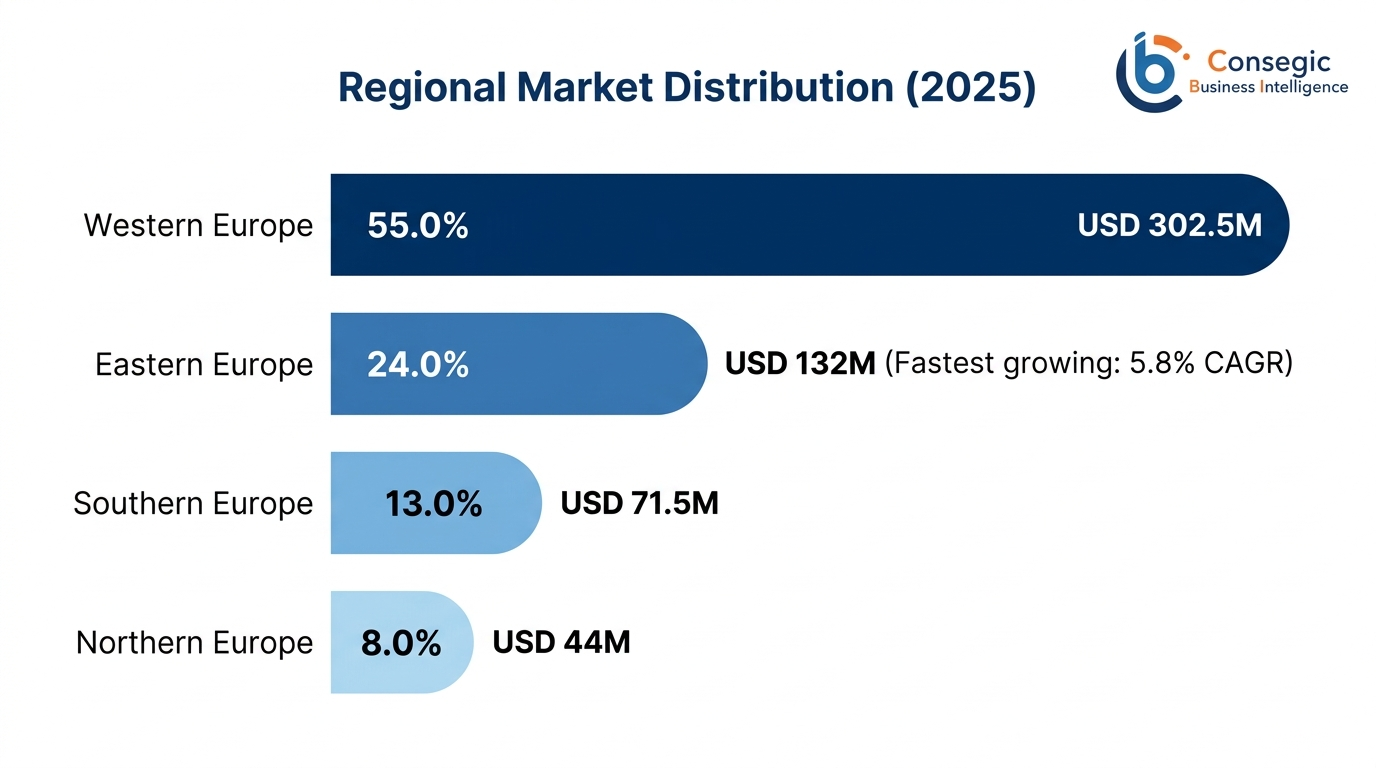
In the Western Europe, the market dominance is USD 302.5 million of market value of 55.0 percent in 2025; this is expected to increase to USD 451 million in 2034. The three countries of Germany, France, and the United Kingdom represent 71 percent of Western European market value; a result of the developed healthcare facilities with a hospital bed density of 6-8 beds per 1,000 population, high volume of surgical procedures amounting to 18 million operations per year across the three countries, and the established clinical guideline that requires a rigorous clinical protocol with mandatory DVT prophylaxis. The EUR 4,000- 6,000 of health spending in healthcare is appropriate to promote the use of advanced technologies and the preference of high-quality products both at the institutional and home level of healthcare.
Western European markets are also dominated by Germany, which has a market share of about 34 percent in the region, which is a good representation of the hospital infrastructure in Germany with 1,900 acute care facilities, a good presence of medical device manufacturing companies including those with well established sequential compression systems, and well-developed reimbursement systems by having statutory health insurance of 88 percent of the population. The compliance of clinical guidelines is 78-85 percent of surgical DVT prophylaxis guidelines, which is pushing the use of uniform compression systems through the hospital networks. Advanced multi-chamber systems and integrated monitoring platforms are particularly strong on the German market, along with adopted digital healthcare using progressive healthcare policies of digitalization of healthcare services, which allows reimbursement of those who use remote patient monitoring services with the quality.
France has 23 percent of the Western European market value and has centralized healthcare system structure with universal coverage, government measures that facilitate surgical safety such as EUR 45 million that has been allocated to hospital quality improvement programs in 2023 and an increasing focus on ambulatory surgery with 63 percent of eligible procedures being performed in outpatient environments. The rates of adopting sequential compression systems are high in the French hospitals with 72 percent of the surgical departments making use of mechanical prophylaxis as a measure of standardized protocols. Expansion of home healthcare segment through the national healthcare strategy of 25 percent conversion of suitable hospital care to home care by 2030 presents a big growth opportunity to the portable compression system that has remote monitoring features.
Eastern Europe: Healthcare Modernization and the fastest growth.
With the highest regional growth potential of USD 132 million of the market value at 24.0 percent in 2025, which will increase to USD 213.2 million by 2034, with the highest CAGR of 5.8 percent based on market value, Eastern Europe appears to be superior to other regional markets. This can be attributed to the extensive modernization of healthcare infrastructures under EU structural funds projects with an EUR 8.5 billion allocation on the modernization of healthcare facilities across the member states throughout the 2021-2027 programming period as surgical volumes grow at 4.2 percent per year as more people gain access to healthcare and more healthcare become aware of the evidence-based approaches to DVT prevention and the continued growth of middle-class populations with increasing healthcare spending power to find the application of premium medical technology.
Poland dominates the markets in Eastern Europe with a regional market share of 32 percent, where the EU accession-driven healthcare system reforms helped it, with hospital modernization programs in the last five years spending EUR 2.1 billion on the upgrades of its facilities and medical equipment acquisition, and expanding the surgical capacity to 2.8 million procedures per year through both the public and the private healthcare system. In 2021, the Polish National Health Fund established standardized DVT prevention measures with necessities to document the use of mechanical prophylaxis thoroughly in the surgical population, establishing systematic institutional demand in the use of compression systems with built-in monitoring and compliance verification features.
Romania and Czech Republic are the two countries that contribute to 28 percent of the market value in Eastern Europe, and they exhibit high healthcare modernization paths. The National Health Strategy 2022-2027 of Romania budgeted EUR 1.8 billion in terms of hospital infrastructure and medical technologies, and specially focused on the acquisition of surgical safety equipment such as advanced compression systems. Czech Republic has good foundations of manufacturing economy and well developed medical device production capabilities where domestic compression system assembly business operations cover the regional markets at a cost competitiveness level. The two nations are showing an increasing trend towards the use of home healthcare delivery model including government initiatives that assist chronic disease management in the outpatient care setting which is establishing an increasing demand on portable compression apparatus with user-friendly working features.
Competitive Landscape and Key Market Players
Strategic Positioning and Market Leadership
Arjo AB (Sweden) - It is the Leader of Comprehensive Vascular Health Solutions.
Arjo holds the profit leadership position with a projected 2024 European revenues of EUR 78-86 million as a market leader of 18-20 percent with a more confident market leader of 25-28 percent in the institutional hospital market segments. The competitive positioning of the company focuses on its broad product portfolio in the family of Flowtron sequential compression system, with advanced multi-chamber devices with 12 independent compression zones, programmable pressure delivery of 30-120 mmHg and with complex microprocessor control to support 200+ programmable compression protocols designed to meet application-specific clinical requirements and target patient populations.
Clinical differentiation is founded on the basis of deep-sea evidence base based on more than 180 published peer-reviewed clinical efficacy studies using the product in a wide range of patient populations, integration capacities with the wider patient handling/hygiene product systems and the well-built long-term relationships with European hospital procurement departments through a combination of product quality and intensive service support. These aspects are highlighted in distribution strategy, which includes direct sales force of about 450 clinical specialists in the continent of Europe, hands-on product demonstration, extensive clinical education ventures that train 35,000+ medical professionals per year and responsive technical services networks whereby 24-48 hrs response rate is seen in maintenance of equipment as well as responding to clinical inquiries.
Cardinal Health (United States - Europe Operations) - Institutional Market Focus.
Cardinal Health achieves considerable presence in Europe at an estimate of 2024 revenues of EUR 52-58 million, which constitutes 12-14 percent of the total market share and concentrated power of 22-25 percent of the large hospital system and group purchasing organization segments. Kendall SCD sequential compression system portfolio offers complete institution level solutions with focus on clinical reliability, standardized protocols and low-cost implementation using the system in a variety of hospital settings. Technical differentiation has fast inflation cycles, standardized pressure profiles to work with large groups of patients, and high clinical construction to withstand intensive clinical applications in high volume facilities.
The commercial strategy capitalizes on the established medical product distribution channels with more than 8,500 health care facilities in Europe, a combination selling strategy that bundles compression systems with extended surgical and critical care product lines and pricing structures that is competitive especially to cost sensitive institutional consumers. In 2024, the company invested EUR 6.2 million in the development of the European market and promoting clinical education, building payer relationships in different European healthcare systems, and improving service infrastructure to systematically implement compression therapy in hospital networks that focus on standard protocols of prophylaxis.
Tactile medical (United States - Europe Operations) - Home Healthcare Specialization.
Tactile Medical has good European market positions with projected 2024 regional revenues of EUR 38-44 million with a 8-10 percent total market share and a dominating 25-30 percent market share in lymphedema management and home healthcare market share. The Flexitouch Plus and Entre systems offer superior pneumatic compression using directional flow technology that is proprietary, and offer compression patterns specifically capable of offering lymphatic drainage application with accurate pressures of 20-60 mmHg in sophisticated 8-chamber systems able to mimic the massage techniques of the hand.
Clinical positioning is focused on the specialization of lymphedema with extensive regulatory approvals such as FDA clearance and CE marking of both primary and secondary lymphedema treatment indications, and with clinical studies which show 65-78 percent limb volume reduction with standardized 12-week treatment programs. The commercial strategy focuses on home health channels via specialized lymphedema care centers, vascular medicine clinics, and direct to patient initiatives with comprehensive reimbursement navigation services that help patients with insurance covers to apply within a range of European healthcare systems with different coverage policies and clinical criteria requirements.
Recent Industry Developments
Digital Transformation and Connected Health Platform Integration (2024-2025).
Major manufacturers such as Arjo, Cardinal Health and Tactile Medical unveiled next-generation sequential compression systems with enhanced connectivity capabilities to meet the healthcare digitalization trends and remote patient monitoring needs. In March 2024, Arjo released Flowtron Connect, which combines cellular connectivity to transmit treatment data in real time to secure cloud-based monitoring dashboards, the capability of automated compliance reporting to raise clinical alerts when a treatment is missed or when treating patients in an inappropriate pattern, and predictive maintenance algorithms to schedule preventive services before equipment failures happen.
It was observed that the platform was successfully implemented in 120 European hospitals in the first 9-month implementation timeframe and that the rate of protocol adherence improved by 32 percent relative to non-connected systems and 18 percent through proactive equipment maintenance scheduling. In September 2024, Cardinal Health announced improved Kendall SCD Smart Compression systems, which included new advanced pressure monitoring, automated patient assessment, and connectivity to hospital electronic health record systems to facilitate the smooth clinical record-keeping and quality assurance tracking in institutional compression therapy programs.
Circular Economy Programs and Sustainability Initiatives (2024-2025).
Large manufacturers developed detailed sustainability strategies in accordance with the European Green Deal goals and the environmental responsibility concerns of the healthcare sector. In June 2024, Arjo made big announcements of carbon reduction commitments with a goal of 50 percent product carbon footprint reduction by 2030 by designing compression garments with recycled polyester and bio-based materials, using packaging that reduces material use by 35 per cent, and device electronics that use less energy by 40 per cent and maintain clinical performance levels.
The company created a very elaborate program of garment recycling that takes used compression sleeves at 250 collection points throughout Europe where recycled materials of non-medical use are made and the medical waste materials are suitably handled. In November 2024, Cardinal Health introduced circular economy initiative, which included systematic take-back program of end-of-life compression systems, refurbishing working units and reselling to cost-sensitive markets at 40-50 percent less than the price of new equipment, and recycling electronic components and housing materials in a systematic way through certified environmental management programs.
(2024-2025) Clinical Evidence Development and Market Access Enhancement.
Manufacturers had made huge investments in the process of clinical evidence that prove the effectiveness of compression therapy, health economics, and increased therapeutic indications. Tactile medical funded multi-centered European clinical trial of 340 lymphedema patients in Germany, France, and Italy, comparing advanced pneumatic compression therapy to traditionally used conservative treatment methods, such as manual lymphatic drainage and compression garment treatment. Preliminary results, which were first presented at European Venous Forum in June 2024, showed 43 percent better limb volume reduction with pneumatic compression, 52 percent better quality-of-life assessment score, and a good cost-fold ratio of EUR 12,400 per quality-adjusted life year gained in submitting reimbursement applications in countries where coverage policy is currently limited.
Arjo worked with European health care economics research consortium that has studied in detail the real-world evidence results of DVT prevention in 85 hospitals in 8 countries with systematic mechanical prophylaxis and pharmacological-only programs in matched patient populations. Results of studies published in December 2024 showed that the incidence of symptomatic DVT had decreased by 38 percent with the implementation of mechanical compression, 45 percent with the implementation of a mechanical compression to reduce the incidence of pulmonary embolisms, and a net saving of EUR 1,850 per surgical patient in healthcare costs through complication prevention and shorter length of stay.
Europe Sequential Compression System Market Report Insights
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2022–2034 |
| Base Year | 2025 |
| Forecast Period | 2026–2034 |
| Market Size in 2024 | USD 520 Million |
| Market Size in 2025 | USD 550 Million |
| Market Size in 2034 | USD 820 Million |
| CAGR (2026–2034) | 4.5% |
| By Product Type | Multi-Chamber Sequential (60.0%), Single-Chamber Uniform (24.0%), Portable Home-Use (11.0%), Specialized Clinical (5.0%) |
| By Application | DVT Prevention (45.0%), Lymphedema Management (35.0%), Chronic Venous Insufficiency (20.0%) |
| By Distribution Channel | Medical Equipment Distributors (55.0%), Direct Hospital Sales (26.0%), Home Healthcare Distributors (12.0%), Online Platforms (7.0%) |
| By Region | Western Europe (55.0%), Eastern Europe (24.0%), Southern Europe (13.0%), Northern Europe (8.0%) |
| Key Players | Arjo AB, Cardinal Health, Tactile Medical, Zimmer Biomet, DJO Global, Mego Afek, Bio Compression Systems |
| Report Coverage |
|
Key Questions Answered in the Report
What is the size, and forecast, of the market of Europe sequential compression system? +
The European market of sequential compression system exhibits low growth traits of USD 550 million in 2025 and USD 820 million in 2034 respectively, which is a growth rate of 4.5 percent per annum within the forecast duration. The maturity in the market differs by region in Europe as the Western Europe has established adoption patterns whereas the Eastern Europe has demonstrated heightened growth prospects due to the healthcare modernization plans. The industry has a target installed base of 85,000-95,000 compression units in hospital, rehabilitation and home healthcare facilities, and equipment sales volume of 12,000-14,000 units/year with huge recurrent garment and accessory revenues to maintain the growth in the market.
What are the main stimulant factors that are fueling the growth of markets? +
The growth driver force will focus on the ageing demographic profile in Europe whereby the age 65 years and above are presently 20.8 percent of the population in 2023 and the population is projected to increase to 29.4 percent in 2050, directly related to increased risk of venous thromboembolism among the age group because of this age bracket is 10-15 times higher than the demographic groups below this age group. This is owing to rising volumes of surgical procedures of more than 32.4 million operations in Europe each year and clinical guidelines that require mechanical compression as the prophylaxis of DVT among moderate and high-risk surgical patients. The shift towards home healthcare delivery models with annual home healthcare spending of EUR 68 billion increasing at 6.8 percent broadens the range of addressable market opportunities in the institutional setting to chronic disease treatment applications involving long-term compression therapy protocols.
What are the differences between adoption and growth potential in the regional markets of Europe? +
The presence of major regional differences typifies the European sequential compression market, and this is due to the differences in healthcare system organizations, level of economic development, and the standardization of clinical practices. Western Europe controls 55.0 percent market share and developed adoption trends, willing to buy expensive products and has extensive reimbursement systems to support its institutional and home health use. Western European share of Germany (34 percent) is leading the market in the region due to strong hospital infrastructure, solid presence of medical devices industry in the country, and progressive policies of digital health reimbursement. Eastern Europe exhibits the highest growth rate at 5.8 percent CAGR due to the EU structural funding of EUR 8.5 billion on healthcare modernization, growth in surgical volumes, and growth in middle-class healthcare spending in favour of the adoption of advanced medical technology in the region.
What competitive forces dominate the European market of the sequential compression systems? +
European market has moderate competitive structure involving the three major manufacturers sharing about 42-46 percent of the market and the rest of the market was shared by specialized regional players and niche manufacturers. Arjo has a market leadership of 18-20 percent share, which she utilizes through complete product portfolios, developed relationships with hospitals, and over decades of experience in the market through developed infrastructure distribution throughout Europe. The level of competition in different market segments is quite different as institutional markets focus on clinical evidence, full-service infrastructure, and developed procurement relations, whereas home healthcare markets are characterized by convenience, patient-related comfort, and remote monitoring options. Entry level competition is greater and in most clinical applications, clinical efficacy and total cost of ownership issues would support highly multi-chamber systems.
What are the emerging technological trends in future product development and markets? +
Major technological progress in the area is on connectivity and digital health integration, where connected compression systems have the ability to include cellular communication options, cloud-based patient management systems, and mobile application interfaces that allow whole body remote patient management and compliance adherence. These features will solve the most important issues of treatment adherence, increasing compliance to therapy by traditional levels of 45-60 percent with the use of traditional devices to 75-85 percent with connected devices due to automated patient reminders, provider monitoring capabilities, and interaction features provided to patients. Other areas of innovation are miniaturization and portability improvements that allow the expansion of home healthcare, pressure management advances such as microprocessor-based devices providing accurate multi-chamber compression patterns, and improvements to the user interface such as touchscreen controls and simplified operation protocols that allow patients to self-administer their medication with little clinical training.