Home > > Pharmaceuticals > > Human Growth Hormone Market Size, Trends, & Industry Outlook (2035)
Human Growth Hormone Drugs Market - Size, Share, Industry Trends, and Forecasts (2025-2035)
ID : CBI_3451 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
Human Growth Hormone Drugs Market Scope & Overview
The global human growth hormone (HGH) drugs market is a niche and highly controlled market segment of the biopharmaceutical industry that includes recombinant DNA-derived somatropin formulations and novel growth hormone analogs long-acting growth hormone long-acting growth hormone analogs that have been developed to treat growth hormone deficiency (GHD) in children and adolescents, short stature diseases such as Turner syndrome, Prader-Willi syndrome, chronic kidney failure, idiopathic short stature, small for gestational age (SGA The market can be assessed as USD 5,850 Million in 2024, and USD 6,200 Million in 2025 (base year), and projected to grow immensely to USD 11,500 Million by 2035. This growth trend reflects a compound annual growth rate (CAGR) of 6.4 per cent over the forecast period of 2026 to 2035 due to revolutionary formulations of long-acting growth hormones that cut down injection frequency to weekly or monthly dosages, higher diagnosis rates of growth hormone deficiency through clinical awareness creation and standardised diagnostic guidelines, more approved indications than classic GHD, more recognition of adult growth hormone deficiency as a legitimate medical condition requiring treatment, biosimilar market growth that enhances affordability and access to treatment, and large emerging market growth
Regional Trends and Insights
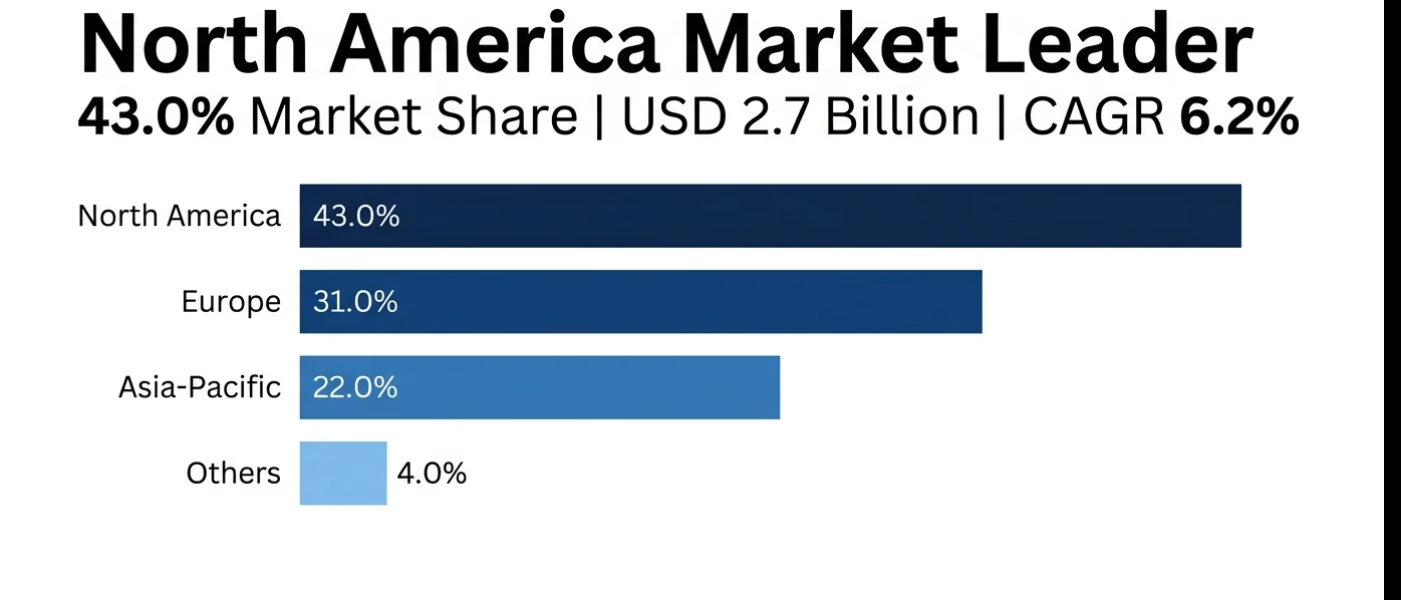
- In 2025, the North American human growth hormone drugs market is USD 2,666 million (43% of global market) and is projected to reach USD 4,830 million by 2035 at a 6.2% CAGR.
- In 2025, the European human growth hormone drugs market is USD 1,922 million (31% of global market) and is projected to reach USD 3,450 million by 2035 at a 6.0% CAGR.
The 191-amino-acid polypeptide, human growth hormone, is naturally secreted by the anterior pituitary gland, and has important physiological functions in the whole life such as the stimulation of linear growth in childhood, regulation of protein synthesis, promotion of lipolysis, regulation of glucose metabolism, bone mineralization, maintenance of muscle mass, and maintenance of organ functioning. Growth hormone deficiency is caused by congenital genetic mutations, pituitary tumors, cranial irradiation, traumatic brain injury, or idiopathic conditions, and is characterized by severely retarded growth velocity in children (usually less than 4 cm/year), reduced adult height potential, abnormal body composition (with an increased level of adiposity), diminished bone mineral density, poor quality of life, and metabolic dysfunction (such as dyslipidemia, insulin resistance).
In 1985, the FDA approved recombinant human growth hormone (rhGH) therapy, which transformed therapy with the replacement of cadaveric HGH derived in the pituitary, which is known to cause Creutzfeldt-Jakob disease. Current rhGH cell lines exhibit the same amino acid sequence as the endogenous somatropin with strict purification protocols that produce pharmaceutical grade product of high specifications in terms of potency, purity and safety.
The total global treatment population comprises of approximately 285,000 pediatric patients, 175,000 adult patients with growth hormone therapy, and the addressable market represents an estimated 1.8-2.2 million undiagnosed or untreated patients worldwide which is a huge growth opportunity with the increased awareness of the need to treat and a wider access to such treatment in emerging markets.
Market Channel Distribution Analysis
The market of human growth hormone drugs is offered by four separate channels, which address the needs of select healthcare delivery systems with divergent regulatory frameworks, reimbursement models and patient access systems. The overall market share of revenue has a high degree of dispersion according to healthcare system structure and geographical market features:
Distribution Channel Failure (2025):
- Hospital Pharmacies and Specialty Clinics: USD 2,728 Million (44.0% of the total market value)
- Retail Pharmacies and Specialty Pharmacy Networks: USD 2,046 Million (33.0% of market revenue)
- Online Pharmacies and Direct to Pat Distribution: USD 930 Million (15.0% of market share)
- Other Channels (Compounding Pharmacies, Institutional Sales): USD 496 Million (8.0% of the total market value)
Investment in human growth hormone research and development, and long-acting formulation development like the development of once-weekly long-acting growth hormone formulations with TransCon technology or PEGylation technologies, digital health integration, connected injection devices to provide adherence cues, biosimilar development to lower the cost of treatment, and clinical research expanding approved indications in metabolic disorders and niche patient populations all reached USD 1,850 Million in 2024, and major biopharmaceutical companies allocate about 18-22% of
The industry is undergoing paradigm shift to patient-focused delivery models solution to the problem of low treatment adherence with studies showing that 30-45% of children with a multi-year dose of medication, which averages 4-8 years, display suboptimal adherence to daily injection schedules, needle phobia, and lifestyle disruption, and fatigue with treatment.
Human Growth Hormone Drugs Market Dynamics (DRO)
Key Drivers:
Radical Growth Hormone Formulations Revolution.
The greatest innovation force in therapy has been the most efficient productions and commercialization of long acting growth hormone preparations which have changed the treatment paradigm of daily subcutaneous injections to weekly or monthly schedules of administration plan. Sogroya (somapacitan) by Novo Nordisk has generated reversible albumin binding technology that prolongs half-life of daily rhGH (2-4 hours) to about 3 days in order to provide sustained therapeutic concentrations with once-per-week dosing.
The TransCon hGH (lonapegsomatropin) of Ascendis Pharma is based on Transient Conjugation (TransCon) technology that temporarily conjugates growth hormone to polyethylene glycol carrier using cleavable linker to form prodrug that is released slowly to give sustained physiologic exposure. Non-inferior height velocity outcomes were shown during clinical trials against daily rhGH and significant quality-of-life benefits were seen with 86% fewer injections.
According to the patient preference studies, 78-85 percent of pediatric patients and caregivers indicated a preference of weekly to daily injections, and the adherence rate to daily regimens was 65-75 percent compared to the 88-95 percent adherence rate to weekly formulations in clinical trials, which translates into better clinical outcomes since the growth response is strongly associated with adherence to therapy.
Raising Rates of Diagnosis and Clinical Awareness.
Significant market expansion comes on the basis of the increase in the rate of diagnosis of growth hormone deficiency due to the spread of clinical awareness, the establishment of diagnostic guidelines, and the increase in the number of endocrinology specialists. Traditionally, the deficiency of growth hormone was underdiagnosed to a large extent, and it was estimated that not more than one-fourth to one-third of children with treatable growth disorders are properly examined and treated.
Pediatric growth hormone deficiency occurs in 1:4,000-1:10,000 of children and adult growth hormone deficiency in 1:10,000-1:2,000 adults with childhood onset disease, and 1:10,000 adults with adult onset disease caused by pituitary tumors, surgery, radiation, or traumatic brain injury.
Connected Devices and Digital Health Integration.
A huge opportunity provided by the integration of digital health tools is the smart injection pens that track the data of dosing and connect with mobile apps, letting the parents and physicians keep the eye on adherence in real-time. Real-time monitoring and patient engagement capabilities associated with connected devices have been shown to significantly improve adherence over conventional devices by between 15-20% and gamification features used with pediatric patients have further led to greater levels of compliance.
Key Restraints:
Expensive Treatment and Reimbursement Problems.
The highest market access barrier is still the high price of growth hormone treatment with the cost of treatment as USD 15,000-40,000 annually depending on the weight of the patient, the choice of the product and the geographical prices of the market. The reimbursement approval will involve a substantial amount of paperwork to prove medical necessity by showing confirmed deficiency of growth hormone by conducting stimulation tests, recorded growth failure, and delay of bone maturation with insurance companies requiring strict prior authorization and continuous monitoring guidelines.
Literature suggests that 1/5 to 1/4 of patients requiring clinical growth hormone deficiency are denied reimbursement or face delayed payments, which becomes an obstacle to access especially in those markets with minimal insurance or high cost-sharing requirements.
Health and Safety Issues and Investigations.
Although the growth hormone therapy shows an overall good safety profile when used correctly, the effects have the potential of causing adverse effects, which pose prescribing caution and regulatory challenges. The concerns are documented in terms of elevated intracranial pressure, slipped capital femur epiphysis, changes in glucose metabolism with the possible development of insulin resistance and theoretical issues concerning the development of cancer especially in patients with malignancy history.
Because of its possible use as an abused drug in sports and anti-aging markets, hGH is a regulated drug in numerous jurisdictions with regulatory authorities mandating Risk Evaluation and Mitigation Strategy (REMS) programs and comprehensive post-marketing surveillance research.
Biosimilar Competition/Price Erosion.
The blockbuster daily hGH drugs patent expired, and the market is now opened to biosimilars that trade at 20-30% discount to reference drugs, further increasing the price competition in the daily injection category, and biosimilars are supporting volume growth by increasing access to treatment and affordability.
Future Opportunities:
New Delivery Systems and Non-injectable Formulations.
There is a high innovation potential with respect to the creation of oral or other non-injection delivery systems. Though growth hormone has a large molecular size which poses a problem to oral bioavailability, new technologies such as permeation enhancers, nanoparticle encapsulation, transdermal microneedle patches, and intranasal delivery systems show promising preclinically positive results in overcoming basic adherence issues.
Emerging Market Expansion
There is a significant growth potential in emerging markets such as China, India, Brazil and the Southeast Asian countries whereby there will be better healthcare systems, endocrinology specialists will increase in number, more people will have health cover and the middle-class will be rising to give a good opportunity to expand. The market of growth hormones in China has an outstanding growth rate of 15-18% CAGR due to the cultural focus on height and widening of the reach of the private insurance coverage.
Biomarker Development and Precision Medicine.
Combining genetic analysis, predictive modeling based on baseline auxology, IGF-1 measurements, and compliance records would assist in determining which children would benefit optimally on particular regimens, reasons to continue therapy and reasons to withdraw, and to offer evidence of value-based contracting strategies to payers.
Market Segmentation Analysis
By Product Type: Formulation Analysis
Daily Growth Hormone Injections: Foundation in the Market.
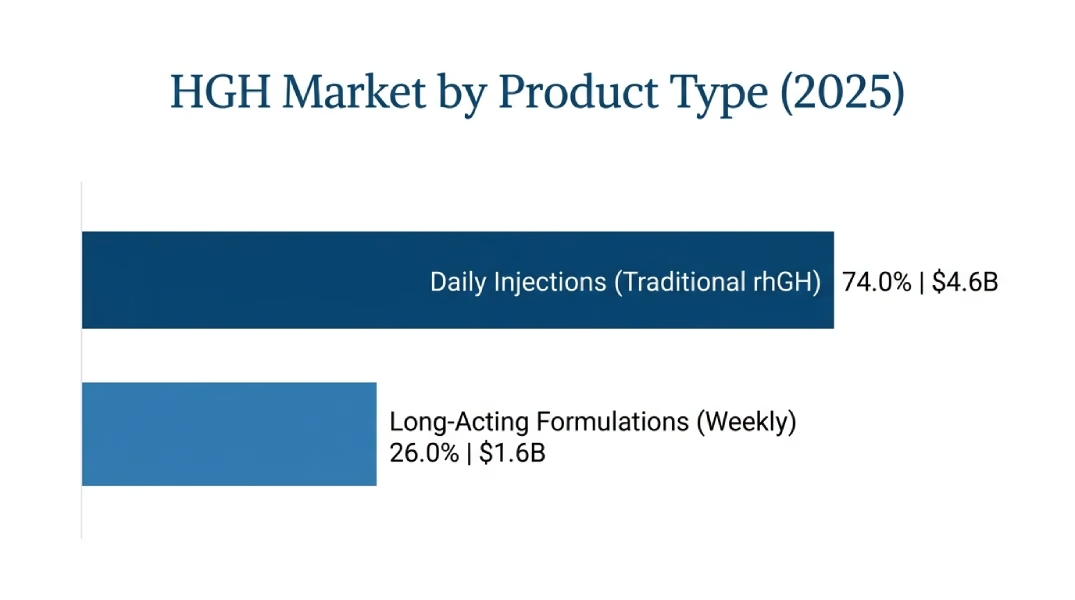
The daily recombinant human growth hormone preparations remain in the market at USD 4,588 Million or 74.0% of total market value in the year 2025, and are expected to increase to USD 8,050 Million of in the year 2035 at a 5.8% CAGR. Traditional rhGH preparations that have to be used daily as a subcutaneous injection system are included in this segment including Genotropin (Pfizer), Norditropin (Novo Nordisk), Humatrope (Eli Lilly), Saizen (Merck KGaA), and a variety of biosimilar products.
Long-Acting Growth Hormone Formulations: Ground Breaking Innovation.
LAGF formulations have USD 1,612 Million (26.0% of market value), the rapidly expanding market with 9.5 CAGR due to the superior levels of compliance and preference by the patients. Sogroya (somapacitan-beco, Novo Nordisk) on adults with GHD and TransCon hGH (lonapegsomatropin-tcgd, Ascendis Pharma) on children with GHD are also part of this segment, but other products are in late-stage development that is establishing a competitive base.
Through Application: Indication Analysis.
Pediatric Growth Hormone Deficiency: Maximum Sign.
Pediatric growth hormone deficiency USD 3,224 Million (52.0% of total market value) includes children who have known GHD, due to provocative testing and growth failure. The therapy is usually maintained until close to the final height or when the bone is fully age consistent with results achieved following years of therapy with great success provided that the treatment commences at an early age.
Adult Growth Hormone Deficiency: Increasing Recognition.
The USD 1,364 Million (22.0% of market value) represents the adult growth hormone deficiency, which is an expanding indication because of the increasing acceptance of adult GHD as a valid medical condition requiring treatment due to body composition changes, low quality of life, and metabolism disorders.
Other Approved Indications
The rest of the approved indications such as Turner syndrome, Prader-Willi syndrome, chronic kidney disease, idiopathic short stature, and SGA are USD 1,612 Million (26.0% of market value) in total.
Regional Market Analysis
North America: Innovation Powerhouse and Leadership in the market.
The market for North American human growth hormone drugs will have USD 2,666 Million in 2025, which will be 43.0 percent of market value globally, and will increase to USD 4,830 Million by 2035 at a CAGR of 6.2 percent. The United States is the biggest regional consumer with 92.0 percent of market value with a high level of infrastructure of endocrinology specialists, insurance coverage of all indications approved, and diagnostic abilities and regulatory environment promoting innovation.
The U.S market is the best user of treatment per capita in the world as 140000 pediatric patients and 85000 adult patients are estimated to have been treated with growth hormone therapy, which indicates excellent rates of diagnosis and access to treatment than foreign markets.
Europe: Biosimilar Leadership and Universal Healthcare.
By the year 2025, the European market of human growth hormone drugs is worth USD 1,922 Million worth 31.0% of the global market value with forecasts reflecting that the market will grow to USD 3,450 Million by 2035 at a CAGR of 6.0%. Germany is the market share of 24.0 percent of the market value of the region and the market shares of France, United Kingdom, and Italy are substantial.
The European market shows the greatest biosimilar penetration in the world with 35-45 market share in the country as Europeans have pioneered biosimilar regulatory framework and the national healthcare system focusing on the cost-effective treatment modalities.
Asia-Pacific: The Quickest Market.
The Asia-Pacific market of the human growth hormone drugs will have USD 1,364 Million market value by 2025 with a solid increase but lower now, at USD 2,645 Million by 2035 with 6.8 CAGR as the fastest growth rate in the region. China leads the regional demand with 52.0 percent of market value portraying tremendous growth rates which are good due to cultural focus on height, growing middle-income, rising healthcare facilities, and competition among manufacturers within the country.
Japan has 24.0% of Asia-Pacific market value with mature market attributes, whereas South Korea makes 12.0 percent with good growth pace due to competitive domestic manufactures and cultural aspects that focus on height.
Other Regions
Latin America has USD 186 Million (3.0% of world market), Middle East and Africa have USD 62 Million (1.0%), both having growth prospects as the healthcare infrastructure is built and the supply of specialists is raised.
Competitive Landscape and Key Market Players
Market Leadership and Strategic Positioning
Novo Nordisk A/S (Denmark) - Innovation and Market Leader.
The market leadership of Novo Nordisk with its estimated 2024 growth hormone revenue of USD 1,450-1,600 Million, which currently constitutes an estimated 24-26% global market share, is due to its total portfolio which includes Norditropin (daily rhGH) and Sogroya (once-weekly somapacitan). First-mover advantage in long-acting growth hormone, advanced delivery devices and extensive support programs given to patients are the elements that contribute to the competitive advantage of the company.
Pfizer Inc. (United States) - Assured Market Structure.
Pfizer has about 20-22 percent market share in the world with an estimated 2024 growth hormone revenue of USD 1,250-1,400 Million on Genotropin brand family and has a strong standing with its overall dosing solutions, new delivery systems and strategic alliances with OPKO Health in long-acting formulations.
Aspirin Pharma A/S (Denmark) - Disruptor of Innovations.
TransCon hGH (lonapegsomatropin) by Ascendis has changed the market playing on proprietary technology TransCon that uses sustained release formulations. The company is only in long-acting premium segment that is experiencing good growth in the pediatric usage.
Other Market Leading Industry Parties:
- Eli Lilly and Company (United States) - Humatrope having robust market in the United States.
- Merck KGaA (Germany) - Saizen with advanced systems of delivery with interconnected devices.
- Novartis (Switzerland): Omnitrope biosimilar leadership in Europe.
- Changchun GeneScience (China) - Jintropin leadership in the Chinese market.
- Ferring Pharmaceuticals (Switzerland) Zomacton niche positioning.
Recent Industry Developments
Long-Acting Growth Hormone Commercial Expansion (2024-2025).
TransCon hGH of Ascendis Pharma has been available throughout the expansion markets in Europe after it was first approved in the United States with clinical results showing no difference in height velocity and significant quality-of-life benefits with weekly injections. The product received high market acceptance and growth path.
Digital Health Integration and Connected Devices (2024-2025)
The most prominent manufacturers introduced interconnected delivery devices and mobile applications with adherence monitoring, reminder to inject, growth tracking, and telemedicine integration, where the system demonstrated an increase in adherence by 15-20% over the traditional devices due to real-time monitoring and patient interaction features.
Biosimilar Market Expansion (2024-2025).
Various biosimilar companies had increased geographic coverage and product range, regional manufacturers introduced products in new markets at 25-40% lower prices than originator prices, opening access opportunities and stiffening competition.
Human Growth Hormone Drugs Market Report Insights
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2022–2035 |
| Base Year | 2025 |
| Forecast Period | 2026–2035 |
| Market Size in 2025 | USD 6,200 Million |
| Market Size in 2035 | USD 11,500 Million |
| CAGR (2026–2035) | 6.4% |
| By Product Type | Daily Injections (74.0%), Long-Acting Formulations (26.0%) |
| By Application | Pediatric GHD (52.0%), Adult GHD (22.0%), Other Indications (26.0%) |
| By Distribution | Hospital Pharmacies (44.0%), Retail/Specialty (33.0%), Online (15.0%), Others (8.0%) |
| By Region | Asia-Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Players | Novo Nordisk, Pfizer, Ascendis Pharma, Eli Lilly, Merck KGaA, Sandoz, Changchun GeneScience, Ferring |
| Report Coverage |
|
Key Questions Answered in the Report
What is the size of Human Growth Hormone Drugs market? +
The global human growth hormone drugs market has a strong growth potential with the USD 6,200 Million in 2025 and projected to reach USD 11,500 Million by 2035 and with 6.4 recurring growth rate demonstrates sustained growth due to revolutionary formulations of growth hormone, once every week rather than every day, improves treatment compliance rates up to 88-95 percent, and has a large addressable market size of 1.8-2.2 million undiagnosed or untreated individuals worldwide.
What is the most booming area of the Human Growth Hormone Drugs market? +
Asia-Pacific is the most rapidly growing regional market with an annualized growth of 6.8% CAGR, with China as the biggest contributor to the growth with its outstanding growth at the expense of height, as a measure of social and economic prosperity, growing middle-class populations with disposable income to spend on healthcare, improving healthcare infrastructures and the availability of experts in endocrinology, domestic manufacturers compete with each other and offer their products at competitive prices, and overall regional growth of the economy supporting the growth in healthcare spending. Nonetheless, North America is a market leader at 43.0% market share because of advanced endocrinology facilities, extensive insurance penetration, the highest per capita treatment rates in the world, and the regulatory system that encourages innovation such as approval of long-acting formulations by FDA.
How are the key drivers of market growth to 2035? +
Notable growth drivers are the long-acting growth hormone formulations that turned the treatment paradigm upside down with weekly administration lowering injection burden by 86 percent and adherence by 65-75 percent to 88-95 percent, and increased the rate of diagnosis by increasing clinical awareness with traditionally 25-35 percent of all children with treatable growth disorders receiving appropriate evaluation, the improved level of addressability with increasing population of addressable patients with classic GHD, turner syndrome, prader-willi syndrome, and idiopathic short stature.
What obstacles inhibit market expansion? +
The main issues are high treatment costs of USD 15,000-40,000/year resulting in a significant financial burden, reimbursement difficulties with 18-25 percent of patients experiencing insurance rejections or delays, stricter prior authorization and compels excessive documentation of medical necessity through provocative testing and growth failure documentation, safety issues such as intracranial pressure risks, glucose metabolism changes, and hypothetical cancer risks necessitating continuous monitoring, regulatory scrutiny due to potential abuse in sports and anti-aging products.
What are the future innovation opportunities to the year 2035? +
Significant opportunities include further development of long-acting formulations with several candidates in late-stage development developing competitive space, new delivery technologies such as transdermal microneedle patches, oral formulations with permeation enhancers, and intranasal delivery systems overcoming the barriers of adherence to injection, biosimilar market expansion offering cost-effective alternatives supporting access in the price-sensitive market, digital health integration with connected devices, mobile apps, telemedicine platforms, and AI-assisted growth prediction algorithms maximizing treatment outcomes, penetrating new markets with bettering healthcare infrastructure.
