Home > > Pharmaceuticals > > Open System Isolator Market Size, Share, Trends & Growth Analysis Report - 2032
Open System Isolator Market - Size, Share, Industry Trends, and Forecasts (2025 - 2032)
ID : CBI_1993 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
Open System Isolator Market Size:
Open System Isolator Market size is growing with a CAGR of 6.3% during the forecast period (2025-2032), and the market is projected to be valued at USD 14,253.03 Million by 2032 from USD 8,820.00 Million in 2024.
Open System Isolator Market Scope & Overview:
An isolator is a containment device that utilizes barrier technology for the enclosure of a controlled workspace. An open system isolator is a self-contained unit that allows materials to enter and exit continuously or semi-continuously through openings while keeping contamination out. Openings in these isolators are engineered using continuous overpressure to exclude the entry of external contaminants into the isolator.
Types of open system isolators including containment isolators, aseptic isolators, and others are employed for aseptic procedures that require products to be constantly entering and exiting the workspace. These isolators serve crucial roles in the applications of aseptic filling, sterile compounding, hazardous drug handling, aseptic transfer, and others making them critical components for end users such as pharmaceutical and biotechnology companies and contract manufacturing organizations. Regulatory mandates in the pharmaceutical sector, growing adoption of biologics, and utilization of these isolators in the production of advanced therapy medicinal products are some of the prominent factors supporting the open system isolator market growth.
How is AI Impacting the Open System Isolator Market?
AI is revolutionizing the open system isolator market by integrating robotic systems to automate critical, high-precision tasks like material transfer and aseptic filling. This reduces human intervention, significantly lowering the risk of contamination and human error. AI-driven sensors and real-time monitoring systems provide continuous environmental data, enabling predictive maintenance and proactive issue resolution before they impact sterility. By analyzing vast datasets, AI optimizes processes, enhances quality control, and ensures stringent regulatory compliance, leading to increased efficiency, safety, and productivity for pharmaceutical and biotechnology manufacturers.
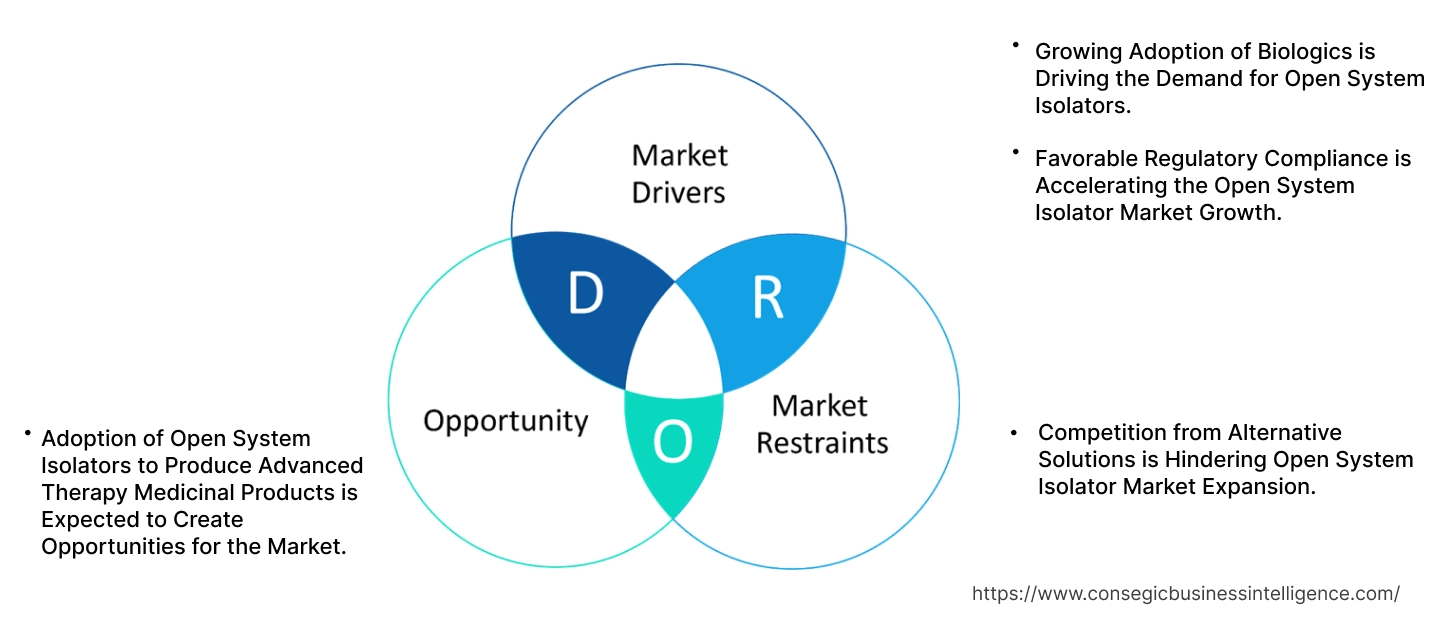
Open System Isolator Market Dynamics - (DRO) :
Key Drivers:
Growing Adoption of Biologics is Driving the Demand for Open System Isolators.
Biologics is a class of medicines derived from living organisms, such as bacteria, yeast, or mammalian cells. They are complex and often used to treat serious conditions like cancer, autoimmune diseases, and infectious diseases. These complex therapies, often used to treat chronic diseases like cancer and autoimmune disorders, demand stringent manufacturing processes to ensure their safety and efficacy. Biologics are highly sensitive to contamination, making it crucial to maintain a sterile environment during their production. Open system isolators provide a controlled environment that minimizes the risk of contamination, making them suitable for handling biologics. As a result, they are being increasingly adopted in the production of biologics.
- For instance, in November 2021, WuXi Biologics, a global company with leading open-access biologics technology platforms, announced it has successfully launched the GMP operation of its new drug product facility located in Wuxi, China, which is the eighth operational drug product facility in the global network of WuXi Biologics. The 12,000m² drug product facility, called DP2, features a state-of-the-art isolator filling line for biologics. This advanced equipment ensures the highest standards of quality and safety in the production of these complex medicines.
Overall, the growing adoption of biologics is creating a necessity for these isolators in the pharmaceutical and biotechnology sectors.
Favorable Regulatory Compliance is Accelerating the Open System Isolator Market Growth.
The open system isolator market expansion is significantly influenced by favorable regulatory compliance. Stringent regulatory requirements across various regions, such as those outlined by agencies like the FDA (Food and Drug Administration) in the United States and the EMA (European Medicines Agency) in Europe, emphasize the importance of aseptic processing, contamination control, and operator safety. These regulations mandate the use of advanced containment technologies like open system isolators to ensure product sterility, minimize contamination risks, and protect both workers and the environment.
Furthermore, regulatory bodies are increasingly focusing on the implementation and adherence to Good Manufacturing Practices (GMPs). Compliance with these GMPs necessitates the use of technologies that ensure product quality, safety, and consistency.
- For instance, the European Union’s Annex 1 good manufacturing practices (GMP) for manufacturing sterile medicinal products mandates the use of isolators as the manufacture of sterile products is subject to special requirements in order to minimize risks of microbial, particulate, and pyrogen contamination.
Therefore, favorable regulatory compliance acts as a driver for the open system isolator market analysis, compelling manufacturers and healthcare providers to adopt these technologies to ensure compliance with stringent regulations and maintain the highest standards of quality and safety.
Key Restraints:
Competition from Alternative Solutions is Hindering Open System Isolator Market Expansion.
Open system isolators face competition from alternative containment technologies including closed system isolators. Restricted Access Barrier Systems (RABS), and biosafety cabinets among others impact their adoption. RABS is a type of restricted access barrier system for the aseptic processing of pharmaceutical products that reduces or eliminates interventions into the critical zone high level of asepsis. RABS offers a less restrictive environment compared to isolators. They allow for greater flexibility in material handling and process operations, potentially reducing operational costs. In terms of investment and operational costs, RABS offers the advantage of a low investment.
Closed isolator systems exclude external contamination of the isolator’s interior by accomplishing material transfer via an aseptic connection to auxiliary equipment, rather than the use of openings to the surrounding environment. Closed systems remain sealed throughout operations offering the highest level of sterility. The availability of these alternatives influences the adoption of these isolators, potentially limiting their market growth.
Future Opportunities :
Adoption of Open System Isolators to Produce Advanced Therapy Medicinal Products is Expected to Create Opportunities for the Market.
Advanced therapy medicinal products (ATMPs) are a class of innovative therapies that include gene therapy, cell therapy, and tissue engineering. These therapies offer transformative treatments for various diseases, including cancer, genetic disorders, and autoimmune diseases. ATMPs pose specific manufacturing challenges beyond those typically addressed by pharmaceutical chemistry. They are highly sensitive to contamination and require precise handling. Hence, there is a growing demand to improve quality assurance and efficiency in the production of ATMPs. Open system isolators provide a controlled environment that minimizes the risk of contamination, making them appropriate for the production of these complex therapies.
- For instance, according to the data published by the Parenteral Drug Association in May 2024, it is stated that isolator technology plays a vital role in optimizing operations for ATMP production further improving product safety, and integrity, and consistently meeting the regulatory requirements. This highlights the benefits offered by isolator systems including for ATMPs.
As a result, the combination of the increasing need for ATMPs, their critical need for controlled manufacturing environments, and the growing recognition of isolator technology's benefits strongly suggest a promising open system isolator market opportunity.
Open System Isolator Market Segmental Analysis :
By Product Type:
Based on product type, the market is categorized into containment isolator, aseptic isolator, and others.
Trends in the Product Type:
- Development of specialized isolators for specific applications such as those used in the handling of radioactive materials, biological agents, and nanomaterials.
The aseptic isolator segment accounted for the largest market share in 2024.
- Aseptic isolators are engineered enclosure systems designed to establish a sterile and isolated environment for the execution of aseptic processes and the manufacture of sterile products.
- They are designed to ensure complete and continuous separation between the internal environment of the isolator and the external room environment, including personnel.
- These systems incorporate overpressure and HEPA (High-Efficiency Particulate Air) filtration to maintain air purity within the chamber. Furthermore, the interior of the isolator is routinely decontaminated using validated methods, typically involving vaporized hydrogen peroxide or other approved decontamination agents.
- Aseptic isolators find widespread application in various pharmaceutical manufacturing processes. These include aseptic filling and finishing, which encompasses activities such as filling, sealing, and labeling sterile drug products. They are also utilized in sterile compounding and the preparation of sterile medications in hospitals and pharmacies.
- They significantly reduce the risk of microbial contamination during critical manufacturing processes. By maintaining a sterile environment, they ensure the sterility and integrity of pharmaceutical products, enhancing patient safety. Moreover, they enhance worker safety by protecting operators from exposure to hazardous materials. Finally, aseptic isolators contribute to increased efficiency by streamlining aseptic processes and providing a controlled and efficient working environment.
- Overall, aseptic isolators are critical for ensuring the safety and quality of sterile pharmaceuticals by providing a controlled, sterile environment, enhancing efficiency, and protecting both products and personnel.
The other segment is expected to grow at the fastest CAGR over the forecast period.
- The others segment includes hybrid isolators.
- Hybrid isolators are designed to provide both aseptic and containment features.
- Aseptic containment isolators (ACIs) are specialized enclosures that provide a high level of both aseptic processing and operator safety during the handling of hazardous materials.
- They uniquely combine aseptic processing capabilities with robust containment features, making them suitable for handling potent compounds or highly hazardous materials while ensuring product sterility.
- ACIs protect both the operator and the environment from exposure to hazardous drugs or other potentially harmful substances. Advanced features such as HEPA filtration and specialized transfer ports minimize the release of hazardous materials into the surrounding environment.
- The benefits of using ACIs include significantly reduced operator exposure to hazardous materials, improved product quality, minimized environmental impact, and increased efficiency in aseptic processes. As a result, there is a growing focus on the adoption of hybrid isolators.
- For instance, in March 2024, Telstar, a company specializing in the development of solutions for aseptic production, launched a new dual-mode isolator system. This new system is capable of operation for both containment and aseptic purposes.
- Overall, as per analysis, hybrid isolators are gaining adoption due to their enhanced safety, improved product quality, and streamlined aseptic processes.
By Application:
Based on application, the market is categorized into aseptic filling, sterile compounding, hazardous drug handling, aseptic transfer, sterility testing, and others.
Trends in Application:
- There is a growing trend towards continuous processing in aseptic filling, which improves efficiency and reduces the risk of contamination.
The aseptic filling segment accounted for the largest market share in the year 2024.
- Aseptic filling is a critical process in pharmaceutical manufacturing, encompassing the sterile filling of liquid or lyophilized drug products into containers such as vials, syringes, and ampoules. Open system isolators play a vital role in this process by providing a controlled and sterile environment that minimizes the risk of contamination.
- The adoption of these isolators for the aseptic filling market is experiencing a significant trajectory driven by the increasing need for injectable drugs and biologics.
- Additionally, technological advancements, such as improved material transfer systems, automated filling systems, and the integration of advanced process analytical technologies are continuously enhancing the efficiency and effectiveness of aseptic filling within open isolator environments.
- For instance, in September 2024, Simtra Biopharma Solutions announced a sterile filling line expansion. The new 150,000 sq. ft. building houses two high-speed automated isolator syringe fill lines and a new high-speed isolator vial line. This supports the adoption of these isolators in aseptic filling.
- Overall, the aseptic filling is witnessing significant growth in the adoption of these isolators, driven by increasing demand, technological advancements, and investments.
The hazardous drug handling segment is expected to grow at the fastest CAGR over the forecast period.
- The rising awareness of the risks associated with handling hazardous drugs, such as cytotoxic drugs and chemotherapeutic agents, is driving the adoption of open system isolators in hospitals, pharmacies, and pharmaceutical manufacturing facilities.
- These isolators provide a safe and controlled environment for healthcare professionals and minimize the risk of exposure to these hazardous substances.
- These isolators act as primary containment barriers, effectively trapping hazardous drug aerosols and preventing their release into the surrounding environment.
- Several factors are driving the increased adoption of these isolators in hazardous drug handling. Growing awareness among healthcare professionals and regulatory bodies regarding the risks associated with handling these drugs is a significant driver.
- The implementation of stricter regulations for the safe handling and disposal of hazardous drugs further emphasizes the need for effective containment solutions. Additionally, an increased focus on worker safety and health in healthcare settings has increased the adoption of these isolators.
- Overall, as per analysis, the rising awareness of risks, stricter regulations, and a strong focus on worker safety are driving the increased adoption of these isolators for the safe and controlled handling of hazardous drugs.
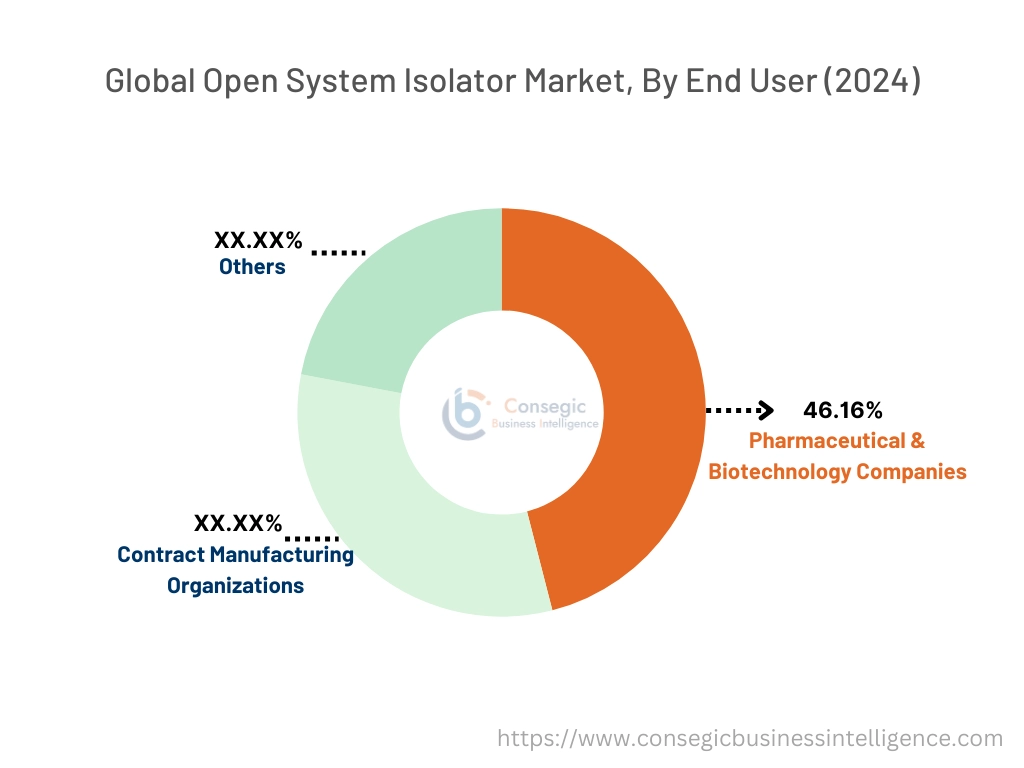
By End-User:
Based on end user, the market is categorized into pharmaceutical & biotechnology companies, contract manufacturing organizations, and others.
Trends in the End User:
- Adoption of advanced technologies such as artificial intelligence and machine learning to optimize isolator operations.
- Increasing investments in high-throughput technologies to increase production capacity to meet the growing demand for biologics and other complex therapies.
The pharmaceutical & biotechnology companies segment accounted for the largest open system isolator market share of 46.16% in the year 2024.
- Pharmaceutical and biotechnology companies constitute the dominant end-user segment. These companies heavily rely on isolators for a wide range of critical operations.
- A primary application is aseptic filling, encompassing the sterile filling where open system isolators provide a controlled and sterile environment, minimizing the risk of contamination.
- Furthermore, these isolators are essential for handling potent APIs (Active Pharmaceutical Ingredients), including cytotoxic drugs and high-potency APIs, providing a safe and contained environment for operators.
- In the production of biologics, such as monoclonal antibodies, vaccines, and gene therapies, isolators play a vital role in maintaining sterility and preventing contamination during critical stages of cell culture, fermentation, and downstream processing.
- Several key factors are driving the trajectory of these isolators within the pharmaceutical and biotechnology sectors. Stringent regulatory requirements regarding aseptic processing, contamination control, and operator safety necessitate the adoption of advanced containment solutions.
- Moreover, pharmaceutical companies prioritize the production of high-quality and safe products, and the isolators play a crucial role in ensuring product sterility and minimizing the risk of contamination.
- Overall, as per analysis, open system isolators contribute to enhanced product quality supporting their adoption within the pharmaceutical and biotechnology sectors.
The others segment is expected to grow at the fastest CAGR over the forecast period.
- Others segment includes hospitals, pharmacies, and research and development organizations.
- Hospitals and pharmacies constitute a significant portion of this segment, utilizing isolators for critical functions such as sterile compounding, preparing sterile medications like intravenous solutions and chemotherapy drugs, and safely handling hazardous medications. The adoption of open system isolators in hospitals is driven by the imperative for safe and efficient handling of hazardous drugs in healthcare settings.
- For instance, in May 2022, Esco Thailand, one of the key players in the open system isolate market, announced the installation of Isoclean Healthcare Platform Isolator at the pharmacy compounding facility of Vibharam Hospital in Bangkok Thailand. The installed HPI unit is a two-glove compounding aseptic containment isolator (CACI) intended for the handling and compounding of chemotherapy drugs for cancer patients.
- Furthermore, research institutions extensively utilize these isolators for various purposes including conducting research on new drug formulations, developing novel therapies, and conducting pre-clinical studies. They are also essential for handling sensitive materials, such as biological materials and radioactive substances, within research laboratories.
- The growing emphasis on research and development activities across the pharmaceutical and biotechnology sectors fuels the requirement for isolators within this segment, supporting scientific advancements and the development of new therapies.
Regional Analysis:
The regional segment includes North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America.
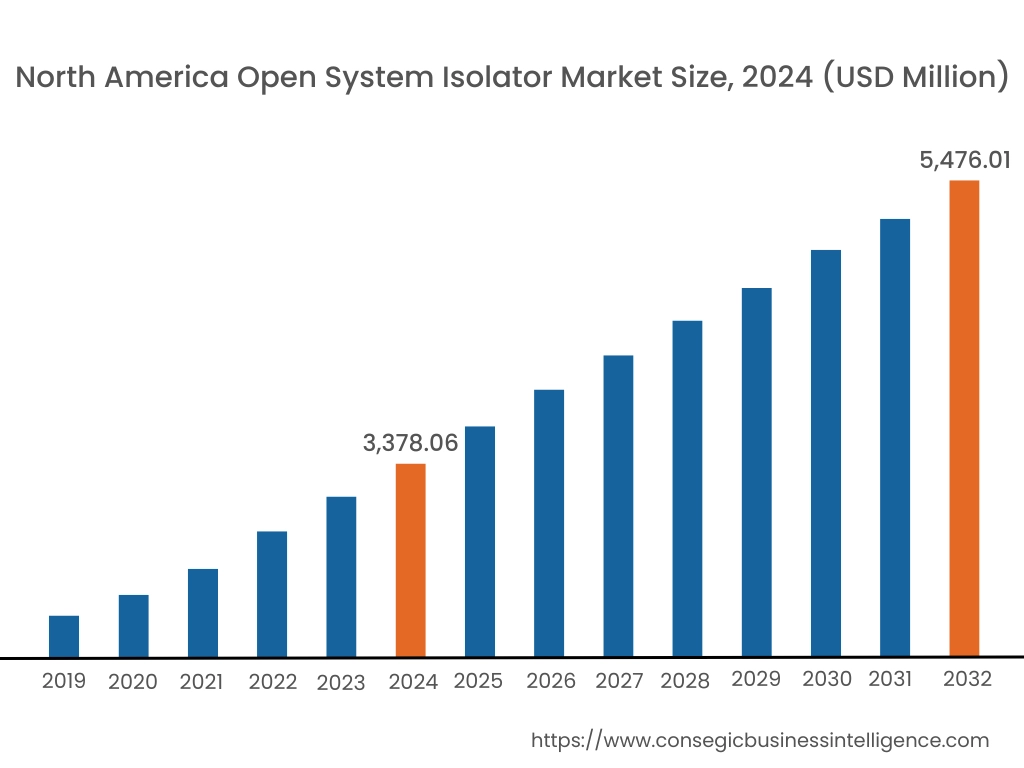
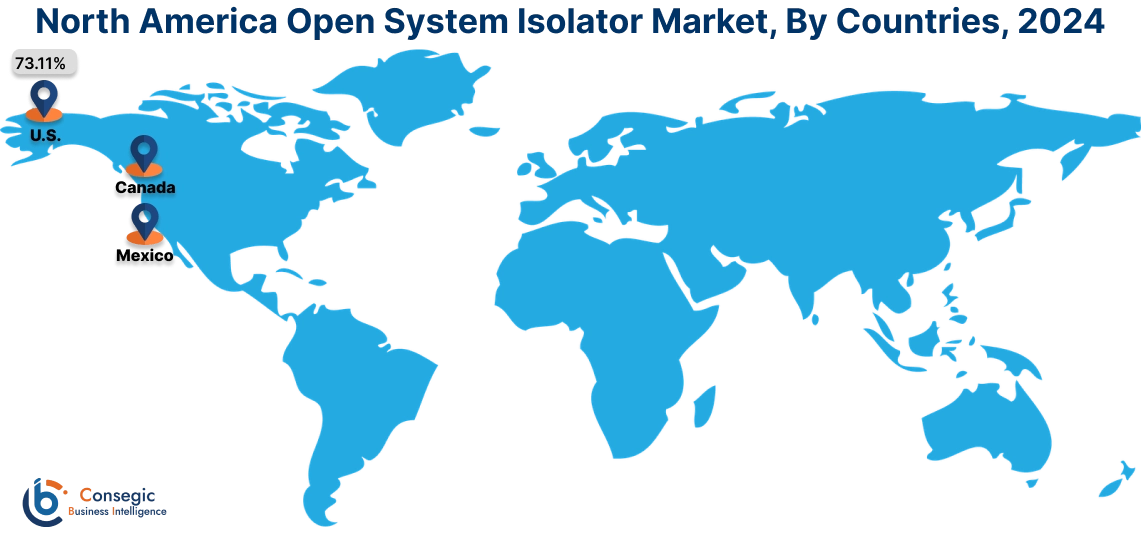
In 2024, North America accounted for the highest market share at 38.30% and was valued at USD 3,378.06 Million and is expected to reach USD 5,476.01 Million in 2032. In North America, the U.S. accounted for the highest market share of 73.11% during the base year of 2024.
North America dominates the global open system isolator market, primarily due to the significant presence of established pharmaceutical and biotechnology companies, driving the requirement for advanced manufacturing technologies, including open system isolators. Additionally, North America serves as a center for pharmaceutical and biotechnology research and development, driving the requirement for advanced technologies and fostering innovation in isolator design and functionality.
- For instance, according to the data published by Pharmaceutical Research and Manufacturers of America in July 2022, it is stated that PhRMA member companies’ R&D investments reached a record high of USD 102.3 billion in 2021.
Furthermore, North America has stringent regulatory frameworks for pharmaceutical manufacturing, emphasizing product quality, safety, and compliance. These regulations drive the adoption of advanced containment technologies such the isolators to ensure adherence to safety and quality standards. Moreover, well-developed healthcare infrastructure, including advanced hospitals and research facilities, provides a strong foundation for the adoption and utilization of sophisticated technologies like isolators. Thus, as per analysis, a combination of the aforementioned factors and trends is driving a substantial trajectory in North America.
Asia Pacific is experiencing the fastest growth with a CAGR of 6.9% over the forecast period. The open system isolator market trend across the region is attributed to a combination of factors including rapid economic growth, a burgeoning pharmaceutical and biotechnology sector, increasing healthcare expenditure, and a growing focus on patient safety and regulatory compliance. This region boasts a rapidly expanding pharmaceutical and biotechnology sector, with significant investments in research and development, manufacturing facilities, and infrastructure. This creates a need for advanced technologies such as open system isolators to ensure product quality, safety, and efficiency. Additionally, ongoing improvements in healthcare infrastructure, including the development of modern hospitals, clinics, and research facilities, are creating a more conducive environment for the adoption of advanced technologies.
Europe presents a well-established position in open system isolator market analysis. Europe boasts a well-established pharmaceutical and biotechnology sector with a strong focus on research and development. This robust sector base creates a significant requirement for open system isolators for various applications, including aseptic filling, handling of hazardous drugs, and the production of biologics. Additionally, Europe has stringent regulatory frameworks for pharmaceutical manufacturing and healthcare practices, emphasizing safety and quality. These regulations drive the adoption of advanced containment solutions like isolators to ensure compliance and maintain high product quality standards. Collectively, these factors create a favorable environment for the open system isolator market demand in Europe.
The Middle East and Africa (MEA) region is witnessing notable open system isolator market demand characterized by significant potential. The region is witnessing a growing pharmaceutical sector with increasing investments in manufacturing facilities, research and development, and infrastructure. This drives the need for advanced technologies like open system isolators to ensure product quality, safety, and compliance with international standards. Moreover, rising healthcare expenditure across the region is fueling the requirement for improved healthcare infrastructure and access to advanced medical technologies, creating a favorable environment for the adoption of these isolators in hospitals, pharmacies, and research institutions. Government initiatives to improve healthcare infrastructure and promote the development of the pharmaceutical sector will further drive market growth. This trend is creating open system isolator market opportunities in the MEA.
Latin America is witnessing steady growth in the open system isolator market share, with significant potential for innovation. The growing pharmaceutical sector, with increasing investments in manufacturing facilities, research and development, and infrastructure, is a key driver. Furthermore, rising healthcare expenditure across many Latin American countries is fueling the requirement for improved healthcare infrastructure and access to advanced medical technologies. This increased investment will create a favorable environment for the adoption of open system isolators in hospitals, pharmacies, and research institutions. Growing awareness of the importance of aseptic processing and the need to maintain sterility in pharmaceutical manufacturing further drive the adoption of these systems. Additionally, increasing emphasis on worker safety and health regulations is driving the requirement for technologies like isolators to protect healthcare workers from exposure to hazardous drugs and other potentially harmful substances. Overall, these factors boost the open system isolator market trend across Latin America.
Top Key Players and Market Share Insights:
The Open System Isolator market is highly competitive with major players providing precise products to the national and international markets. Key players are adopting several strategies in research and development (R&D) and product innovation to hold a strong position in the global Open System Isolator market. Key players in the Open System Isolator industry include-
- Esco Micro Pte. Ltd. (Singapore)
- Syntegon Technology GmbH (Germany)
- NuAire, Inc. (U.S.)
- Germfree (U.S.)
- COMECER S.p.A. (Italy)
- M.A. Industria Macchine Automatiche S.p.A. (Italy)
- Getinge (Sweden)
- TEMA SINERGIE S.p.A (Italy)
- Lancs Industries (U.S.)
- Gelman Singapore (Singapore)
Recent Industry Developments :
Product Launch:
- In March 2024, Telstar, a company specializing in the development of solutions for aseptic production, launched a new dual-mode isolator system. This new system is capable of operation for both containment and aseptic purposes.
Open System Isolator Market Report Insights :
| Report Attributes | Report Details |
| Study Timeline | 2019-2032 |
| Market Size in 2032 | USD 14,253.03 Million |
| CAGR (2025-2032) | 6.3% |
| By Product Type |
|
| By Application |
|
| By End-User |
|
| By Region |
|
| Key Players |
|
| North America | U.S. Canada Mexico |
| Europe | U.K. Germany France Spain Italy Russia Benelux Rest of Europe |
| APAC | China South Korea Japan India Australia ASEAN Rest of Asia-Pacific |
| Middle East and Africa | GCC Turkey South Africa Rest of MEA |
| LATAM | Brazil Argentina Chile Rest of LATAM |
| Report Coverage |
|
Key Questions Answered in the Report
How big is the Open System Isolator market? +
In 2024, the Open System Isolator market is USD 8,820.00 Million.
Which is the fastest-growing region in the Open System Isolator market? +
Asia Pacific is the fastest-growing region in the Open System Isolator market.
What specific segmentation details are covered in the Open System Isolator market? +
Product Type, Application, and End User segmentation details are covered in the Open System Isolator market.
Who are the major players in the Open System Isolator market? +
Esco Micro Pte. Ltd. (Singapore), Syntegon Technology GmbH (Germany), and I.M.A. Industria Macchine Automatiche S.p.A. (Italy) are some of the major players in the market.