Home > > Pharmaceuticals > > Pharmaceutical CRO Market Size Trends, Demand, Outlook & Research 2035
Pharmaceutical CRO Market - Size, Share, Industry Trends, and Forecasts (2025-2035)
ID : CBI_3416 | Updated on : | Author : Yogesh K | Category : Pharmaceuticals
Pharmaceutical CRO Market:
Pharmaceutical CRO Market size is estimated to reach over USD 93.98 Billion by 2035 from a value of USD 42.29 Billion in 2024 and is projected to grow by USD 45.47 Billion in 2025, growing at a CAGR of 7.53% from 2025 to 2035
Pharmaceutical CRO Market Scope & Overview:
Pharmaceutical contract research organizations refer to specialized service providers that support drug development through outsourced research activities. The pharmaceutical CRO industry includes preclinical testing, clinical trial management, regulatory consulting, data management, pharmacovigilance, and laboratory services. The objective is to improve development timelines and manage research costs for pharmaceutical, biotechnology, and medical device companies. The service model supports early-stage discovery programs, late-stage clinical trials, and post-marketing studies across global regulatory markets.
The pharmaceutical CRO market is expanding due to rising R&D outsourcing by drug manufacturers seeking cost control and operational flexibility. Growing clinical trial complexity and stricter regulatory standards are increasing demand for specialized service expertise. Small and mid-sized biotechnology firms are relying on CRO partnerships to access infrastructure without large capital investment. Cross-border trial activity across Asia Pacific and Eastern Europe is also supporting steady revenue growth for global and regional CRO providers.
Pharmaceutical CRO Market Size & Forecast
- 2024 Market Size : USD 42.29 Billion
- 2025 Market Size : USD 45.47 Billion
- 2035 Projected Market Size : USD 93.98 Billion
- CAGR (2025-2035) : 7.53%
- Largest growing country : Asia Pacific
- Fastest growing country : North America
How is the Pharmaceutical CRO Market Market Affected by AI?
AI improves pharmaceutical CRO operations by processing large volumes of clinical trial data, patient records, imaging outputs, and biomarker datasets with higher accuracy and speed. Machine learning algorithms identify protocol deviations, data inconsistencies, and site performance gaps across multi-center trials. This improves monitoring efficiency and reduces manual data reconciliation efforts.
AI systems are also applied in patient recruitment modeling, trial feasibility assessment, and predictive safety monitoring. Algorithms evaluate enrollment patterns, dropout risk, and adverse event signals before regulatory submission. This supports faster decision-making and improves data quality across Phase I to Phase IV studies.
Pharmaceutical CRO Market Dynamics - (DRO):
Key Drivers:
Global increase in chronic disease burden raising drug development activities globally
The rising incidence of oncology, cardiovascular, metabolic, and rare diseases is increasing the volume of drug development programs across global markets. Pharmaceutical and biotechnology companies are expanding clinical pipelines to address unmet medical needs. Higher trial volume is increasing demand for outsourced clinical research, data management, and regulatory support services. CROs are witnessing steady project inflow across Phase I to Phase IV studies.
- For instance, in September 2025, chronic diseases caused 43 million deaths in 2021, accounting for 75% of global non-pandemic deaths. Around 18 million deaths occurred before age 70, with over 80% in low and middle-income countries.
Therefore, the global increase in chronic disease burden is accelerating outsourcing demand in the pharmaceutical CRO market.
Key Restraints:
Regulatory delays across major approval markets restrict project timelines and revenue visibility
Lengthy review cycles and additional data requirements from regulatory authorities are extending approval timelines. Sponsors are revising development strategies and adjusting trial scopes in response to compliance queries. Project delays are affecting milestone-based payments and operational planning for CRO service providers. Revenue predictability remains exposed to regulatory decision timelines across US, Europe, and other key markets.
Thus, regulatory delays across major approval markets are constraining operational efficiency in the pharmaceutical CRO industry.
Future Opportunities:
Expansion of biosimilar and generic drug development programs creates growth avenues
Patent expirations of biologics and branded drugs are increasing development activity in biosimilars and generics. Manufacturers are initiating comparative clinical trials and regulatory filing programs to enter cost-competitive segments. These programs require clinical monitoring, bioequivalence studies, and regulatory documentation support. CROs are expanding service offerings aligned with cost-sensitive development pathways.
- For instance, in November 2025, OCED reported that generics accounted for over 75% of drug volumes in Chile, Germany, the UK, the Netherlands, Canada, New Zealand, and Latvia in 2023. By value, generics averaged 25% of the market and exceeded 50% in Chile and the UK.
Thus, expansion of biosimilar and generic drug development programs is generating additional revenue streams in the pharmaceutical CRO market.
Pharmaceutical CRO Market Segmental Analysis:
By Type:
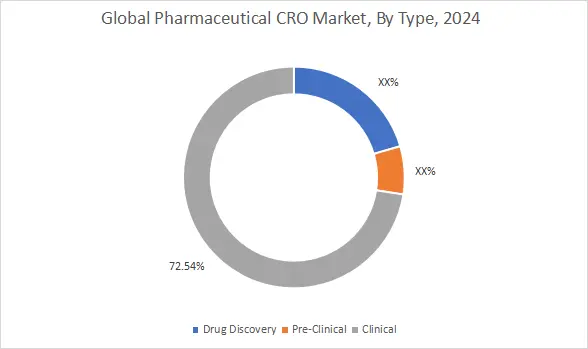
On the basis of type, the pharmaceutical CRO market is segmented into drug discovery, pre-clinical, and clinical.
Trends in the type:
- Increasing outsourcing of early-stage research is expanding demand for integrated discovery services.
- Rising global trial volume is strengthening long-term contracts across clinical phases.
The clinical was responsible for the highest revenue share of 72.54% in 2024.
- Phase III trials involve large patient populations and multi-country operations.
- Moreover, higher protocol complexity increases demand for monitoring and data management.
- In addition, regulatory documentation requirements are higher in late-stage trials.
- Furthermore, milestone-based payments improve contract value.
- Therefore, higher trial scale and longer study duration are supporting dominance of the clinical segment.
It is anticipated that the drug discovery will exhibit the highest compound annual growth rate (CAGR) during the forecast period.
- Biotech startups are increasing early-stage research outsourcing.
- Moreover, virtual discovery models are reducing in-house infrastructure dependence.
- In addition, target validation and lead optimization services require specialized expertise.
- Furthermore, rising innovation in biologics and gene therapies is expanding discovery programs.
- Therefore, expanding early-stage pipelines are expected to drive the drug discovery segment growth.
By Molecule Type:
On the basis of molecule type, the pharmaceutical CRO market is segmented into small molecules and large molecules.
Trends in the Molecule Type:
- Growth in biologics development is increasing demand for specialized testing capabilities.
- Rising complexity of monoclonal antibodies and cell therapies is expanding analytical service requirements.
The small molecules was responsible for the highest revenue share in 2024.
- Small molecules dominate approved drug portfolios.
- Moreover, generic drug development programs rely heavily on small molecule trials.
- In addition, established regulatory pathways support steady project flow.
- Furthermore, bioequivalence studies require structured clinical support.
- Therefore, established development frameworks are supporting dominance of the small molecules segment.
It is anticipated that the large molecules will exhibit the highest compound annual growth rate (CAGR) during the forecast period.
- Biologics pipelines are expanding across oncology and immunology.
- Moreover, complex manufacturing and testing standards require external expertise.
- In addition, biosimilar programs are increasing comparative clinical trials.
- Furthermore, higher per-project value improves revenue realization.
- Therefore, expanding biologics development is expected to drive the large molecules segment growth.
By Services:
On the basis of services, the pharmaceutical CRO market is divided into project management/clinical supply management, data management, regulatory/medical affairs, medical writing, clinical monitoring, quality management/assurance, biostatistics, investigator payments, laboratory, patient and site recruitment, technology, and others.
Trends in the Services:
- Sponsors are preferring bundled service contracts to improve coordination.
- Digital trial platforms are increasing demand for technology-enabled services.
The clinical monitoring was responsible for the highest revenue share in 2024.
- Multi-site trials require structured oversight and compliance tracking.
- Moreover, regulatory inspections require detailed monitoring records.
- In addition, remote monitoring models are increasing operational scope.
- Furthermore, complex protocols increase on-site verification needs.
- Therefore, high operational involvement is supporting dominance of the clinical monitoring segment.
It is anticipated that the data management will exhibit the highest compound annual growth rate (CAGR) during the forecast period.
- Rising trial data volume requires centralized digital systems.
- Moreover, real-time analytics is improving decision timelines.
- In addition, electronic data capture adoption is expanding across regions.
- Furthermore, AI-enabled validation tools are improving data quality control.
- Therefore, increasing data complexity is expected to drive the data management segment growth.
By Therapeutic Areas:
On the basis of Therapeutic Areas, the pharmaceutical CRO market is divided into oncology, CNS disorders, infectious diseases, immunological disorders, cardiovascular disease, respiratory diseases, diabetes, ophthalmology, pain management, and others.
Trends in the Therapeutic Areas:
- Rising cancer incidence is increasing oncology trial volume globally.
- Growing autoimmune and metabolic disorders are expanding specialty pipelines.
Oncology accounted for the largest revenue share in the year 2024.
- Oncology trials involve complex protocols and biomarker-driven designs.
- Moreover, precision medicine approaches require advanced data analytics.
- In addition, multi-regional enrollment increases operational scale.
- Furthermore, higher per-patient trial costs improve contract value.
- Therefore, higher complexity and larger pipeline activity are supporting dominance of the oncology segment.
Immunological Disorders is anticipated to register the fastest CAGR during the forecast period.
- Autoimmune disease prevalence is increasing across developed markets.
- Moreover, biologic and biosimilar programs are expanding clinical pipelines.
- In addition, targeted therapies require specialized monitoring services.
- Furthermore, long-term safety evaluation increases post-marketing studies.
- Therefore, expanding immune therapy research is expected to drive the immunological disorders segment growth.
Regional Analysis:
North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America are the regions of coverage.
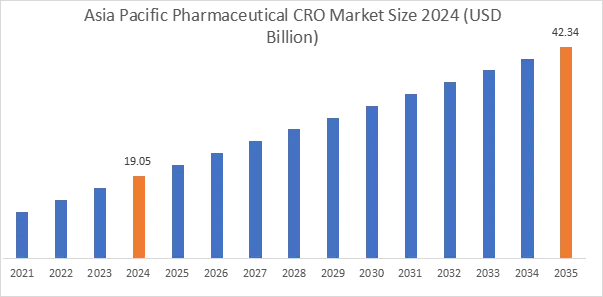
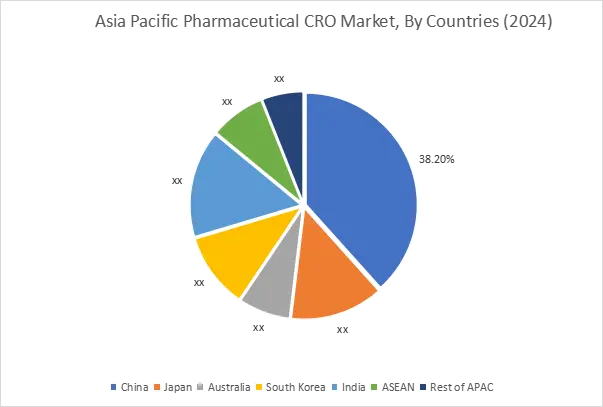
In 2024, Asia Pacific accounted for the highest market share at 45.05% and was valued at USD 19.05 Billion, and is expected to reach USD 42.34 Billion by 2035. China accounted for the highest regional share of 38.2% in 2024. Market growth in the region is supported by cost-competitive clinical trial operations and expanding domestic drug development pipelines. India is strengthening CRO service exports through skilled workforce availability and lower operational costs. In addition, regulatory reforms across China and South Korea are improving approval timelines and cross-border study participation. Growing biologics and biosimilar development programs are sustaining outsourcing growth.
- For example, Southeast Asia recorded 1,146,810 total cancer cases in 2022 as per Lancet Oncology, with 47.6% in men and 52.4% in women, alongside 716,116 deaths (53.8% in men and 46.2% in women).
North America is expected to witness the fastest growth during the forecast period. The US continues to expand oncology and rare disease pipelines, which is increasing late-stage clinical trial activity. Rising adoption of decentralized and technology-enabled trial models is improving operational efficiency. Strong venture funding for biotechnology startups is expanding early-stage research outsourcing. Furthermore, higher regulatory compliance standards are increasing demand for specialized data management and pharmacovigilance services across the region.
- In May 2025, Parexel released a report from a global survey of over 500 pharma, biotech, and CRO leaders/frontline workers, highlighting workforce trends like AI's role in accelerating clinical insights, with 51% seeking AI experts as top hires amid evolving trial complexity.
Europe pharmaceutical CRO market growth is driven by strong clinical research activity in Germany, the UK, and France. Harmonized regulations and expansion of multi-country trials across Central and Eastern Europe are sustaining steady outsourcing demand.
Latin America pharmaceutical CRO market growth is driven by increasing trial participation in Brazil and Mexico. Cost advantages and improving regulatory processes are attracting foreign-sponsored studies.
The Middle East and Africa pharmaceutical CRO market is supported by expanding research infrastructure in the UAE, Saudi Arabia, and South Africa. Government focus on domestic pharmaceutical development is encouraging early-stage clinical activity.
Top Key Players & Market Share Insights:
The pharmaceutical CRO market is moderately consolidated with global full-service providers, mid-sized regional CROs, and niche specialty firms competing across therapeutic segments. Companies are expanding service portfolios through acquisitions and integrated outsourcing models to secure long-term sponsor contracts. Investment in digital trial platforms and AI-enabled data systems is improving operational efficiency and compliance tracking. Expansion of delivery centers in Asia Pacific and Eastern Europe is increasing cost competition across early and late-stage clinical services. Key participants in the market for pharmaceutical CRO include:
- IQVIA Holdings, Inc. – US
- Labcorp Drug Development – US
- Syneos Health – US
- Charles River Laboratories – US
- ICON plc – Ireland
- Parexel International – US
- PPD, Inc. – US
- Medpace Holdings – US
- WuXi AppTec – China
- PSI CRO – Switzerland
Recent Industry Developments:
- In February 2026, Pharmagene Discovery Services relaunched after acquiring a human tissue laboratory near Cambridge, UK, enhancing its CRO capabilities for preclinical and clinical research using consented human tissues.
- In July 2025, Auriga Research partnered with San Francisco Research Institute via MoU to advance AI-integrated global clinical research, regulatory compliance, and wellness product development across India, North America, and Africa.
Pharmaceutical CRO Market Report Insights:
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2019-2035 |
| Market Size in 2035 (USD Billion) | USD 93.98 Billion |
| CAGR (2025-2035) | 7.53% |
| By Type |
|
| By Molecule Type |
|
| By Services |
|
| By Therapeutic Areas |
|
| By Region |
|
| Key Players |
|
| Report Coverage |
|
Key Questions Answered in the Report
How big is the pharmaceutical CRO market? +
The pharmaceutical CRO market size is estimated to reach over USD 93.98 Billion by 2035 from a value of USD 42.29 Billion in 2024 and is projected to grow by USD 45.47 Billion in 2025, growing at a CAGR of 7.53% from 2025 to 2035.
Which segmentation details are covered in the pharmaceutical CRO report? +
The pharmaceutical CRO report includes specific segmentation details for type, molecule type, services, therapeutic areas, and regions.
Which is the fastest segment anticipated to impact the market growth? +
Drug Discovery is the fastest growing segment due to rising early-stage outsourcing.
Who are the major players in the pharmaceutical CRO market? +
The key participants in the pharmaceutical CRO market are IQVIA Holdings, Inc. (US), Labcorp Drug Development (US), Syneos Health (US), Charles River Laboratories (US), ICON plc (Ireland), Parexel International (US), PPD, Inc. (US), Medpace Holdings (US), WuXi AppTec (China), PSI CRO (Switzerland), and others.
What are the key trends in the pharmaceutical CRO market? +
Higher clinical outsourcing, growth in biologics programs, and adoption of AI-based trial tools are shaping the market.
