Home > > Medical Devices > > Point of Care (POC) Diagnostics Market Size, Share & Overview 2035
Point of Care (POC) Diagnostics Market - Size, Share, Industry Trends, and Forecasts - 2035
ID : CBI_3411 | Updated on : | Author : Yogesh K | Category : Medical Devices
Point of Care (POC) Diagnostics Market:
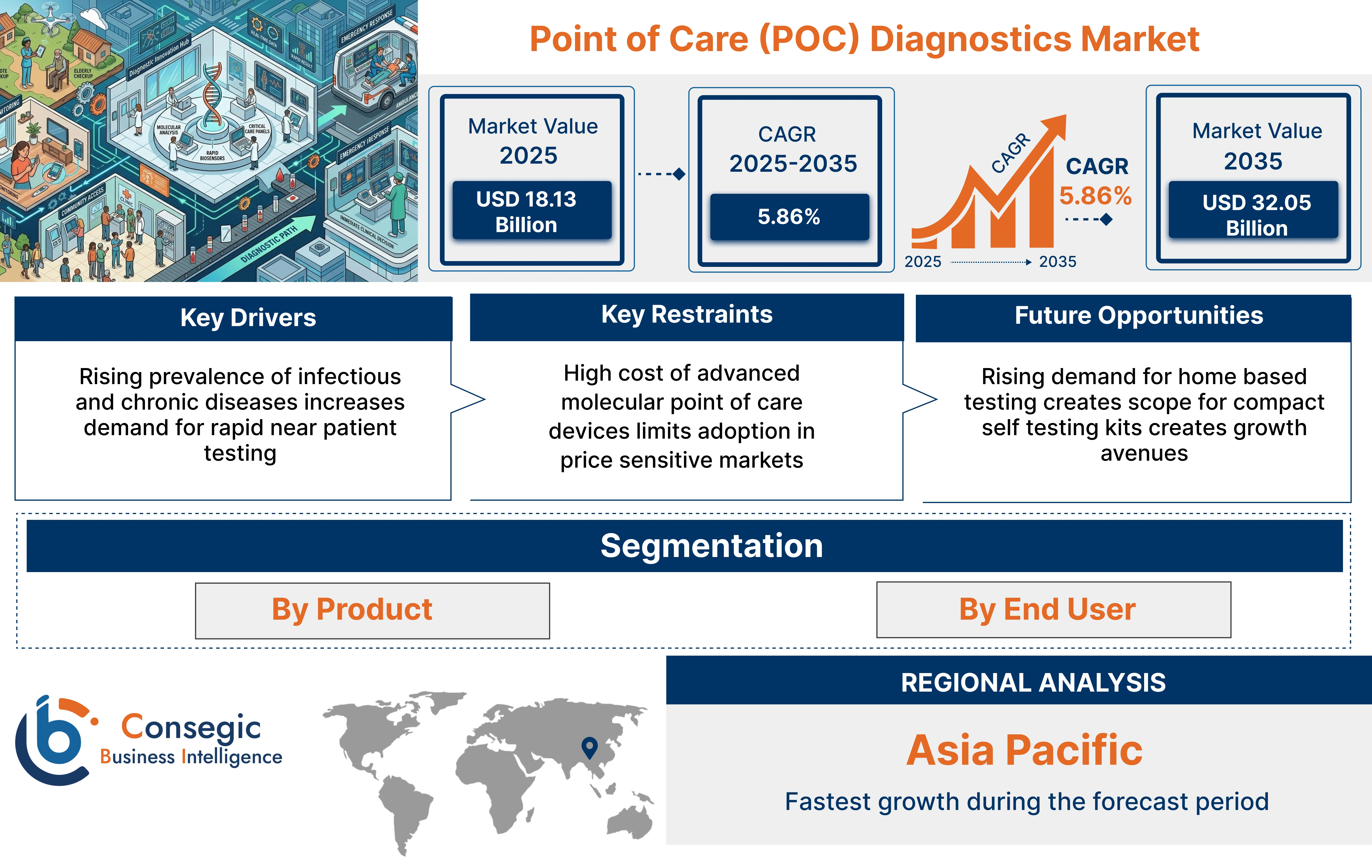
Point of Care (POC) Diagnostics Market size is estimated to reach over USD 32.05 Billion by 2035 from a value of USD 17.13 Billion in 2024 and is projected to grow by USD 18.13 Billion in 2025, growing at a CAGR of 5.86% from 2025 to 2035
Point of Care (POC) Diagnostics Market Scope & Overview:
Point of care diagnostics refers to medical testing conducted near the patient site to enable rapid clinical decision making without reliance on centralized laboratory infrastructure. The industry includes portable diagnostic instruments, rapid test kits, handheld analyzers, molecular testing platforms, biosensors, and connected data management systems. The objective is to deliver timely and accurate results in hospitals, clinics, ambulatory care settings, pharmacies, and home care environments. This includes the detection of infectious diseases, chronic disease monitoring, cardiac marker testing, pregnancy/fertility testing, and emergency care testing within a decentralized healthcare environment.
The near patient testing market is witnessing growth due to the increasing need for rapid testing services and the focus on decentralized healthcare services. The healthcare industry is adopting near patient testing devices as a way to provide quicker services to their patients. The increase in the incidence of chronic diseases and the periodic outbreak of infectious diseases are driving the demand for near patient testing services within developed as well as emerging nations. The integration of digital connectivity and EHR systems is further driving the growth of the near patient testing market.
Market Size & Forecast
- 2024 Market Size : USD 17.13 Billion
- 2025 Market Size : USD 18.13 Billion
- 2035 Evaluate Market Size : USD 32.05 Billion
- CAGR (2025-2035) : 18.13
- Largest growing country : North America
- Fastest growing country : Asia Pacific
How is the Point of Care Diagnostics Market Affected by AI?
Artificial intelligence enhances point of care diagnostics by analyzing clinical data sets, biomarker patterns, and patient history inputs with high precision. Similarly, abnormal test results are detected, early warning signs of diseases are identified, and manual errors are eliminated with the use of machine learning algorithms.
In addition, artificial intelligence systems are integrated with image-based diagnostic tests, molecular diagnostic tests, and connected analytical devices for the purpose of interpreting results and providing risk scores. With the predictive algorithms, the quality control, device performance, and patient data correlations are predicted before the onset of clinical discrepancies, enabling healthcare providers to ensure the accuracy of tests.
Point of Care Diagnostics Market Dynamics - (DRO):
Key Drivers:
Rising prevalence of infectious and chronic diseases increases demand for rapid near patient testing
The incidence of infectious as well as chronic diseases continues to grow at the global level. Early detection of diseases, as well as constant monitoring, facilitates better treatment planning. Point of care diagnostic solutions enable quick results to be obtained at clinics, emergency rooms, as well as community locations. These diagnostic solutions do not require central laboratories. Healthcare providers are increasingly using antigen tests, glucose tests, cardiac marker tests, as well as molecular tests.
- For instance, in September 2025, the WHO states that NCDs resulted in the deaths of 43 million people worldwide in 2021. These deaths were 75% of all non-pandemic-related deaths. In addition, 18 million of these deaths were premature, occurring at the age of 70 or less. These deaths were 82% of all low- and middle-income countries.
Thus, the increasing incidence of infectious as well as chronic diseases is thus creating a greater need for point of care diagnostic solutions.
Key Restraints:
High cost of advanced molecular point of care devices limits adoption in price sensitive markets
Advanced molecular technology involves integrated analyzers, proprietary cartridges, and recurring supplies. Procurement of initial supplies is a financial burden on small hospitals and primary health centers. Budget constraints in developing countries are a challenge to scale up the technology. Reimbursement issues in certain countries are other concerns.
Therefore, the initial cost of advanced molecular technology is a challenge to the scale-up of the technology.
Future Opportunities:
Rising demand for home based testing creates scope for compact self testing kits creates growth avenues
Consumers are looking for convenient options for diagnosis that help reduce the need for hospital visits. Home-use glucose monitors and infectious disease self-test kits are becoming more popular. Manufacturers are launching compact devices that are easy to use and offer digital connectivity options for remote physician access.
- In February 2026, Visby Medical partnered with Watchmaker Genomics to create a home-use test for respiratory pathogen diagnosis using genomic sequencing and POC technology to provide quicker consumer access.
Thus, rising demand for home based testing is creating growth opportunities for compact self testing solutions in the point of care diagnostics market.
Point of Care Diagnostics Market Segmental Analysis:
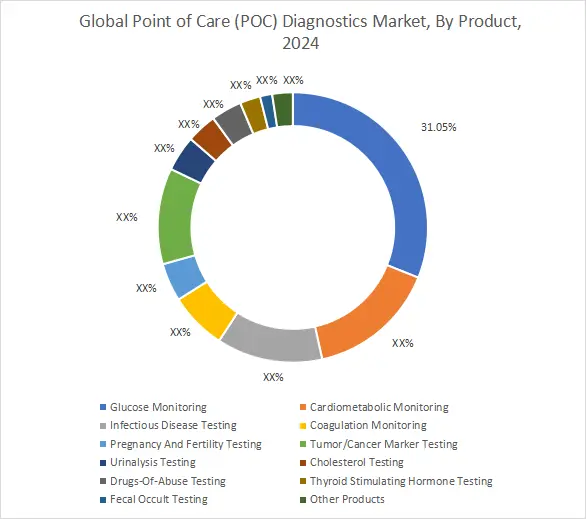
By Product:
On the basis of product, the point of care diagnostics market is segmented into glucose monitoring, cardiometabolic monitoring, infectious disease testing, coagulation monitoring, pregnancy and fertility testing, tumor or cancer marker testing, urinalysis testing, cholesterol testing, drugs of abuse testing, thyroid stimulating hormone testing, fecal occult testing, and other products.
Trends in the Product:
- Recurring infectious disease outbreaks are increasing rapid screening demand.
- Growing focus on early cancer detection is supporting innovation in marker based testing.
The glucose monitoring was responsible for the highest revenue share of 31.05% in 2024.
- Infectious diseases testing includes rapid antigen, molecular, and antibody-based diagnostic kits.
- The high volume of tests conducted across hospitals, clinics, and community health centers drives revenue for the segment.
- Moreover, the ongoing surveillance for respiratory and viral diseases drives routine testing.
- Additionally, the expansion of portable molecular platforms aids diagnostic testing.
- Therefore, sustained demand for rapid infection detection is expected to support the infectious disease testing segment during the forecast period.
It is anticipated that the tumor/cancer marker testing will exhibit the highest compound annual growth rate (CAGR) during the forecast period.
- Tumor/cancer marker testing includes rapid assays for specific oncological biomarkers.
- Rising cancer incidence is increasing demand for early stage screening and monitoring.
- Additionally, the growing demand for personalized treatment strategies drives the development of biomarker-based diagnostic tests.
- Furthermore, the development of miniaturized immunoassay platforms improves the feasibility of the tests.
- In December 2025, IdentifySensors, a company, launches graphene-based check4 sensors for the early detection of cancer, i.e., 50 copies/mL NSCLC, with a detection time of less than 5 minutes, disrupting traditional PCR tests.
- Therefore, growing emphasis on early cancer detection is expected to boost the tumor or cancer marker testing segment during the forecast period.
By End User:
On the basis of end user, the point of care diagnostics market is divided into diagnostic centers, home care, research laboratories, and others.
Trends in the End User:
- Shift toward decentralized healthcare delivery is expanding testing beyond hospital laboratories.
- Increasing patient preference for convenience is supporting home based diagnostics.
Diagnostic centers accounted for the largest revenue share in the year 2024.
- Diagnostic centers perform a wide range of rapid tests across infectious and chronic disease categories.
- Higher patient footfall supports recurring utilization of point of care devices.
- Moreover, structured reimbursement frameworks in urban healthcare systems sustain testing volumes.
- Additionally, availability of trained professionals ensures reliable test interpretation.
- Therefore, strong testing throughput is expected to support the diagnostic centers segment during the forecast period.
Home care is anticipated to register the fastest CAGR during the forecast period.
- Home care includes self testing kits and portable monitoring devices.
- Increasing awareness of preventive healthcare is motivating people to adopt self-monitoring practices.
- In addition, connectivity allows remote physician review of test results.
- Further, expansion of online pharmacy distribution improves product accessibility.
- Therefore, rising consumer adoption of home based diagnostic solutions is expected to boost the home care segment during the forecast period.
Regional Analysis:
North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America are the regions of coverage.
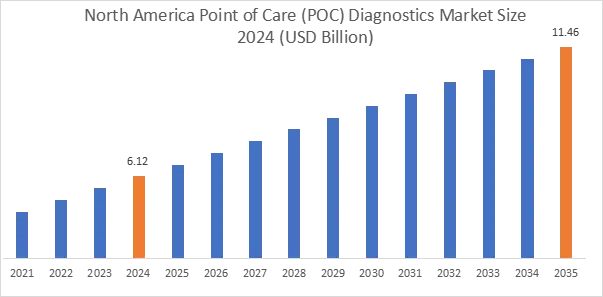
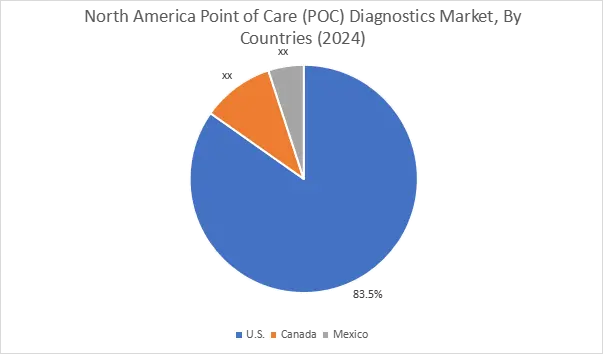
In 2024, North America accounted for the highest market share at 35.75% and was valued at USD 6.12 Billion, and is expected to reach USD 11.46 Billion by 2035. In North America, the U.S. accounted for the highest market share of 83.5% during the base year of 2024. Canada is increasing community healthcare facilities, and this is boosting the use of decentralized testing. Canada is increasing community healthcare facilities, and this is boosting the use of decentralized testing.
- For example, in March 2025, the S. CDC stated that three in four U.S. adults have at least one chronic condition, rising to over 90% for those 65+, 75% for ages 35-64, and 60% for 18-34.
Asia Pacific is expected to witness the fastest growth during the forecast period. China is strengthening primary healthcare networks and local production of rapid test kits. India is expanding infectious disease screening across public health centers. Japan and South Korea are adopting portable molecular platforms in outpatient and elderly care settings. Rising preventive screening awareness is supporting regional demand.
- For example, the Global Fund spends at least USD 600 million annually in India on essential health products like HIV drugs, mosquito nets, and diagnostic kits, with other global entities sourcing cost-effective TB and HIV pharmaceuticals is driving growth in India's POC diagnostics industry through high-volume, affordable supply chains.
Europe market growth is supported by aging demographics and established healthcare systems in the UK, Germany, and France. Routine chronic disease monitoring and structured public healthcare programs are sustaining rapid diagnostic adoption.
Latin America growth is driven by expansion of primary care services in Brazil and Mexico. Public investment in infectious disease surveillance is increasing rapid test deployment.
Middle East and Africa growth is supported by healthcare infrastructure development in the UAE and Saudi Arabia. Screening programs and investments in diagnostic access are increasing the use of point of care testing in hospitals and community facilities.
Top Key Players & Market Share Insights:
The point of care diagnostics market is moderately fragmented with the presence of many global diagnostics players and rapid test manufacturers. Companies are developing their product portfolio for infectious diseases, glucose monitoring, and molecular diagnostics, along with investments in portable devices and connected devices. Partnerships with hospitals, clinics, and pharmacies are also enhancing the distribution footprint for players. Expansion of manufacturing capabilities and multiplexing technologies is also boosting the competitive landscape for players. Key participants in the market for point of care diagnostics include:
- Abbott Laboratories – US
- Hoffmann-La Roche Ltd – Switzerland
- Siemens Healthineers AG – Germany
- Danaher Corporation – US
- Becton, Dickinson and Company – US
- bioMérieux SA – France
- QuidelOrtho Corporation – US
- Sekisui Diagnostics – US
- Trinity Biotech plc – Ireland
- Nova Biomedical – US
Recent Industry Developments:
Product Launches
- In February 2026, Siemens Healthineers worked with World Athletics to use point-of-care blood testing and ultrasound imaging during endurance sports, improving athlete safety and establishing new benchmarks in medical care at sports events.
- In October 2025, Roche highlights how point-of-care testing (POCT) advances like multi-test platforms with microfluidics and AI—enable decentralized healthcare by delivering fast, accurate diagnostics near patients, cutting costs, and improving access in primary care, pharmacies, and remote areas.
- In October 2025, Co-Diagnostics developed a proprietary, low-cost sample prep instrument for its PoC PCR TB test, featuring single-button operation for sputum/tongue swabs, built-in pathogen inactivation, and minimal training needs, with India clinical trials set for year-end.
Point of Care Diagnostics Market Market Report Insights:
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2019-2035 |
| Market Size in 2035 (USD Billion) | USD USD 32.05 Billion |
| CAGR (2025-2035) | 5.86% |
| By Product |
|
| By End User |
|
| By Region |
|
| Key Players |
|
| Report Coverage |
|
Key Questions Answered in the Report
How big is the point of care diagnostics market? +
The point of care diagnostics market size is estimated to reach over USD 32.05 Billion by 2035 from a value of USD 17.13 Billion in 2024 and is projected to grow by USD 18.13 Billion in 2025, growing at a CAGR of 5.86% from 2025 to 2035.
Which segmentation details are covered in the point of care diagnostics report? +
The point of care diagnostics report includes specific segmentation details for product, end user, and regions.
Which is the fastest segment anticipated to impact the market growth? +
Tumor or cancer marker testing is the fastest growing segment, driven by rising demand for early cancer detection and biomarker based screening.
Who are the major players in the point of care diagnostics market? +
The key participants in the point of care diagnostics market are Abbott Laboratories (US), F. Hoffmann-La Roche Ltd (Switzerland), Siemens Healthineers AG (Germany), Danaher Corporation (US), Becton, Dickinson and Company (US), bioMérieux SA (France), QuidelOrtho Corporation (US), Sekisui Diagnostics (US), Trinity Biotech plc (Ireland), Nova Biomedical (US), and others.
What are the key trends in the point of care diagnostics market? +
The market is shaped by rising adoption of rapid infectious disease testing, expansion of home based diagnostic kits, and integration of digital connectivity across portable testing platforms.