Home > > Medical Devices > > Noninvasive Prenatal Testing Market Size, Share, Demand & Analysis (2035)
Noninvasive Prenatal Testing Market - Size, Share, Industry Trends, and Forecasts - (2025 - 2035)
ID : CBI_3421 | Updated on : | Author : Yogesh K | Category : Medical Devices
Noninvasive Prenatal Testing Market Size:
Noninvasive Prenatal Testing Market size is estimated to reach over USD 19.36 Billion by 2035 from a value of USD 5.74 Billion in 2024 and is projected to grow by USD 6.41 Billion in 2025, growing at a CAGR of 11.69% from 2025 to 2035
Noninvasive Prenatal Testing Market Scope & Overview:
The market for non-invasive prenatal testing (NIPT) comprises products and services for testing cell-free fetal DNA in maternal blood for chromosomal abnormalities during pregnancy. These products and services help identify and diagnose chromosomal abnormalities, including trisomy 21, 18, 13, and sex chromosome abnormalities, using next-generation sequencing and data analysis for accurate and timely prenatal screening and testing for healthcare and diagnostic centers.
The market for non-invasive prenatal testing is influenced by increasing maternal age and awareness of early genetic testing for chromosomal abnormalities. Advances in sequencing technology have improved the accuracy of NIPT and decreased testing times. Additionally, NIPT eliminates the need for invasive procedures and reduces miscarriage risk, making it more acceptable for healthcare and diagnostic centers.
Market Size & Forecast
- 2024 Market Size : USD 5.74 Billion
- 2025 Market Size : USD 6.41 Billion
- 2035 Evaluate Market Size : USD 19.36 Billion
- CAGR (2025-2035) : 11.69%
- Largest growing region : North America
- Fastest growing region : Asia Pacific
How is the Noninvasive Prenatal Testing Market Affected by AI?
With the use of AI, the machine learning and bioinformatics tools can be used for the analysis of cell-free fetal DNA sequencing with greater precision and speed. It helps to improve the detection of chromosomal abnormalities and decrease the false positives and false negative results. The AI system also analyzes the genomic data and clinical parameters for better risk assessment.
Noninvasive Prenatal Testing Market Dynamics - (DRO):
Key Drivers:
With the increase in the age of mothers in developed and emerging countries, the risk of chromosomal abnormalities, such as trisomy 21, trisomy 18, and trisomy 13, is also increasing. The mothers who have delayed their pregnancy at an older age are at a greater risk of genetic abnormalities. Therefore, the demand for non-invasive prenatal tests for the early screening of the fetus is increasing. The non-invasive prenatal tests, also known as NIPTs, can be used for the early screening of the fetus and the detection of abnormalities with the use of cell-free DNA analysis.
- For instance, according to data from the Organisation for Economic Co-operation and Development, in all other OECD countries, the average age of women at childbirth is at least 30, and in Greece, Ireland, Italy, Japan, Korea, Luxembourg, Spain, and Switzerland, the average age is above 32.
Therefore, rising maternal age is supporting growth of the noninvasive prenatal testing market.
Key Restraints:
High testing costs restrict accessibility in low and middle-income countries
The cost of non-invasive prenatal testing is also high compared to conventional screening tests. The use of advanced sequencing technology, laboratory facilities, and bioinformatics results in high costs for non-invasive prenatal testing. Lack of reimbursement policies in low and middle-income countries also limits the affordability of these tests.
Therefore, high costs are acting as a restraint for the growth of non-invasive prenatal testing.
Future Opportunities:
Integration of artificial intelligence and bioinformatics platforms enhances genomic data interpretation
The integration of AI and advanced bioinformatics tools in non-invasive prenatal testing is resulting in growth opportunities. The use of AI algorithms in non-invasive prenatal testing improves the interpretation of genomic data and results in high detection accuracy. Machine learning models help in the detection of rare chromosomal abnormalities using massive sequencing data.
- For instance, in June 2025, DeepMind launched AlphaGenome, an AI tool that helps in the better understanding of intricate genomic information by using the tool to predict the impact of genetic variations on cellular function. This took the use of AI in deciphering intricate genomic information to the next level, enabling researchers to gain deeper insights into gene function and disease.
Therefore, integration of artificial intelligence and bioinformatics platforms is expected to boost the noninvasive prenatal testing market in the future.
Noninvasive Prenatal Testing Market Segmental Analysis:
By Gestation Period:
On the basis of gestation period, the noninvasive prenatal testing market is segmented into 0–12 weeks, 13–24 weeks, and 25–36 weeks.
Trends in the Gestation Period:
- The increasing preference for early fetal genetic screening is strengthening demand for testing during the first trimester.
- The growing awareness regarding early detection of chromosomal abnormalities is supporting testing adoption in initial pregnancy stages.
The 0–12 Weeks was responsible for the highest revenue share in 2024.
- Noninvasive prenatal testing is commonly performed during the first trimester to detect chromosomal abnormalities at an early stage.
- Early diagnosis enables informed clinical decisions and appropriate pregnancy management.
- Further, the rising recommendation of first-trimester screening by obstetricians is supporting segment dominance.
- Additionally, improved sensitivity of cell-free DNA testing in early gestation is strengthening utilization rates.
- Thus, increasing emphasis on early risk assessment is driving leadership of the 0–12 weeks segment.
It is anticipated that the 13–24 Weeks will exhibit the highest compound annual growth rate (CAGR) during the forecast period.
- Testing during the second trimester supports confirmatory screening and follow-up assessments.
- The rising number of late prenatal consultations in developing regions is strengthening demand in this timeframe.
- Moreover, the integration of ultrasound and genetic tests in the second trimester of pregnancy is improving diagnostic accuracy.
- In addition, the expansion of access to sophisticated prenatal diagnostic tests is fueling the market segment.
- Therefore, broader screening coverage across pregnancy stages is expected to accelerate growth of the 13–24 weeks segment.
By Pregnancy Risk:
On the basis of pregnancy risk, the market is divided into high & average risk and low risk.
Trends in the Pregnancy Risk:
- The increasing maternal age globally is strengthening demand for genetic screening among high-risk pregnancies.
- The increasing recommendations for universal screening are also helping drive adoption in lower risk populations.
High & average risk represented the highest revenue share in 2024.
- High risk pregnancies are those with advanced age of the mother, abnormal ultrasound results, and family history of genetic disorders.
- Physicians often recommend non-invasive prenatal testing as a primary screening method for these patients.
- Moreover, the likelihood of chromosomal abnormalities in high risk patients is also helping drive testing adoption.
- Insurance reimbursement policies in some countries are also favorable for high risk screening.
- Hence, necessity and medical guidelines are helping drive market dominance in this segment.
Low risk is anticipated to exhibit the most rapid compound annual growth rate (CAGR) throughout the forecast period.
- The expansion of noninvasive prenatal testing as a routine screening option is strengthening adoption among low-risk pregnancies.
- The growing confidence in test accuracy and safety is increasing physician recommendations for broader populations.
- Further, rising patient awareness regarding early fetal health assessment is supporting segment growth.
- Furthermore, healthcare professionals are increasingly adopting universal screening strategies as part of prenatal care protocols.
- Hence, increasing accessibility and expanded guidelines are likely to boost growth of the low-risk market segment.
By Method:
On the basis of method, the noninvasive prenatal testing market is divided into ultrasound detection, biochemical screening tests, and cell-free DNA in maternal plasma tests.
Trends in the Method:
- The shift toward molecular-based screening approaches is strengthening demand for cell-free DNA testing.
- The continued use of traditional screening tools in combination with advanced testing is supporting multi-method diagnostic strategies.
Cell-free DNA in Maternal Plasma Tests accounted for the largest revenue share in the year 2024.
- Cell-free DNA testing involves analyzing DNA fragments from the fetus found in the mother’s bloodstream to screen for chromosomal disorders.
- The superior sensitivity and specificity of this approach are helping drive clinical adoption.
- Furthermore, the reduced risk of complications during testing compared with invasive diagnostic methods is also a factor.
- Additionally, faster turnaround and automation of lab processes are also helping improve efficiency
- Thus, superior diagnostic accuracy is helping drive dominance of the cell-free DNA testing market segment.
Biochemical Screening Tests is anticipated to register the fastest CAGR during the forecast period.
- Biochemical screening tests involve the measurement of biochemical markers in the serum of pregnant women to determine the risk of chromosomal abnormalities.
- The low cost of the test in comparison to molecular screening is encouraging its continued use in resource-constrained settings.
- In addition, the use of biochemical markers in combination with ultrasound findings is improving the use of combined screening tests.
- Further, public healthcare programs are continuing to use serum screening tests as a standard procedure in prenatal care.
- Therefore, affordability and established clinical protocols are supporting sustained adoption.
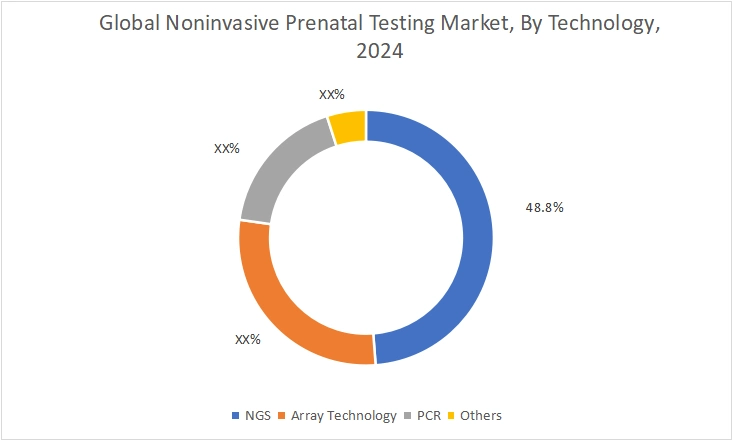
By Technology:
On the basis of technology, the market is divided into NGS, array technology, PCR, and others.
Trends in the Technology:
- The increasing application of high-throughput genomic analysis is strengthening demand for sequencing-based technologies.
- The advancement in molecular diagnostics infrastructure is supporting adoption of precise detection platforms.
NGS represented the largest revenue share of 48.8% in 2024.
- NGS enables comprehensive analysis of fetal chromosomal abnormalities with high accuracy.
- The capability to process multiple samples simultaneously improves laboratory efficiency.
- Furthermore, the rising demand for expanded panels covering microdeletions and rare disorders is strengthening segment dominance.
- Additionally, declining sequencing costs are improving accessibility across diagnostic centers.
- Thus, scalability and analytical precision are driving the leadership of the NGS segment.
PCR is anticipated to register notable growth during the forecast period.
- PCR-based tests can specifically detect certain abnormalities in the chromosomes of a fetus.
- The faster turnaround time will aid in rapid decision-making.
- In addition, the low capital investment in comparison to sequencing machines is encouraging the use of this technology in smaller laboratories.
- Further, continuous technological refinements are improving assay sensitivity.
- Therefore, operational simplicity and cost efficiency are contributing to segment expansion.
By Product:
On the basis of product, the market is segmented into consumables & reagents and instruments.
Trends in the Product:
- The increasing testing volume across diagnostic laboratories is strengthening demand for consumables and reagents.
- The expansion of molecular diagnostic facilities is supporting procurement of advanced testing instruments.
Consumables & Reagents accounted for the highest revenue share in 2024.
- Each noninvasive prenatal test requires reagents, kits, and sample preparation materials, resulting in recurring demand.
- The rising number of prenatal screenings globally is supporting steady product consumption.
- Moreover, the introduction of specialized reagent kits for expanded panels is strengthening segment growth.
- Additionally, laboratory standardization protocols are supporting consistent utilization rates.
- Thus, recurring testing requirements are driving segment dominance.
Instruments are projected to witness stable growth during the forecast period.
- Instruments include sequencing platforms, PCR systems, and sample preparation equipment.
- Expansion of in-house testing facilities in hospitals and laboratories is supporting capital investments.
- Furthermore, the use of automated workflows is improving laboratory efficiency.
- Moreover, technological developments in compact and high-performance equipment are improving efficiency.
- Therefore, infrastructure development and laboratory modernization are contributing to segment expansion.
By Application:
On the basis of application, the noninvasive prenatal testing market is segmented into trisomy, microdeletion syndrome, and other applications.
Trends in the Application:
- The increasing prevalence of chromosomal abnormalities is strengthening demand for targeted screening.
- The expansion of genetic panels is supporting broader clinical applications.
Trisomy held the largest revenue share in 2024.
- Trisomy screening includes detection of conditions such as Down syndrome (Trisomy 21), Trisomy 18, and Trisomy 13.
- The high clinical significance of these disorders is supporting routine screening recommendations.
- Also, the strong accuracy of molecular testing methods is enhancing detection reliability.
- Additionally, public awareness initiatives are increasing acceptance of early trisomy screening.
- Thus, established clinical importance is driving dominance of the trisomy segment.
Microdeletion Syndrome is anticipated to exhibit faster growth during the forecast period.
- Microdeletion testing identifies smaller chromosomal deletions associated with rare genetic conditions.
- The growing adoption of expanded NIPT panels is supporting segment expansion.
- Moreover, the increasing availability of high-resolution sequencing technologies is improving detection capability.
- Further, rising demand for comprehensive prenatal risk assessment is strengthening utilization.
- Therefore, broader genomic analysis is expected to accelerate growth of the microdeletion syndrome segment.
By End-Use:
On the basis of end-use, the market is divided into hospitals & clinics and diagnostic laboratories.
Trends in the End-Use:
- The expansion of prenatal care services is strengthening testing volumes across healthcare settings.
- The centralization of advanced molecular testing is supporting growth of specialized diagnostic laboratories.
Diagnostic Laboratories accounted for the largest revenue share in 2024.
- Laboratories possess advanced sequencing infrastructure and skilled personnel for high-throughput testing.
- The increasing outsourcing of prenatal genetic testing by hospitals is supporting segment dominance.
- In addition, the ability to process large sample volumes efficiently is strengthening operational scalability.
- Further, partnerships between laboratories and obstetric clinics are improving service accessibility.
- Thus, centralized testing capabilities are driving leadership of the diagnostic laboratories segment.
Hospitals & Clinics are projected to register steady growth during the forecast period.
- The growing establishment of in-house molecular diagnostic units is supporting adoption.
- The increasing demand for integrated prenatal care services is strengthening hospital-based testing.
- Moreover, the reduction in turnaround time through onsite sample collection is improving patient convenience.
- Further, expansion of maternal healthcare programs is supporting infrastructure development.
- Therefore, improved healthcare access and service integration are contributing to segment growth.
Regional Analysis:
North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America are the regions of coverage.
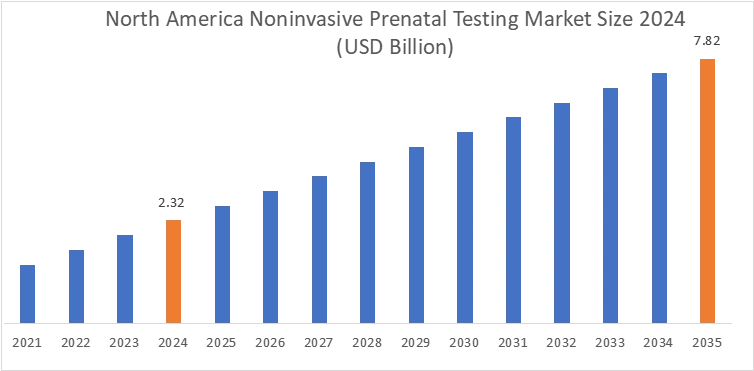
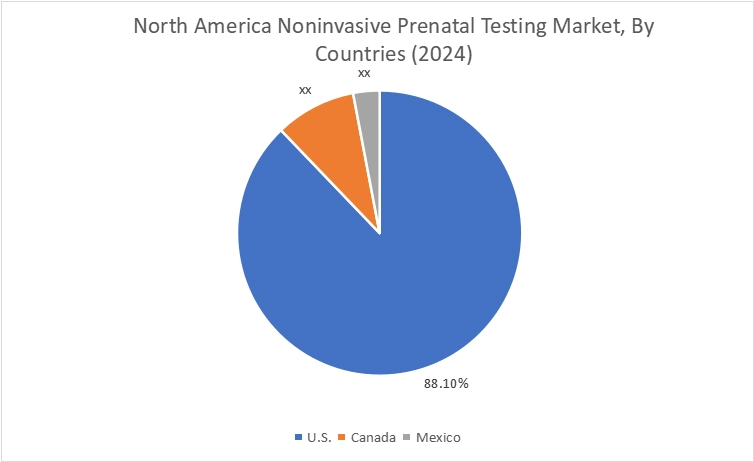
In 2024, North America accounted for the highest market share at 40.37% and was valued at USD 2.32 Billion, and is expected to reach USD 7.82 Billion by 2035. In North America, the U.S. accounted for the highest market share of 88.1% during the base year of 2024. The growth is attributed to the increasing trends in maternal age and high awareness levels regarding early fetal chromosomal screening in the US and Canadian markets. Increasing pregnancy rates among women aged 35 years and above increases the likelihood of chromosomal abnormalities, thus strengthening the noninvasive prenatal testing (NIPT) solution based on cell-free DNA (cfDNA).
- For instance, the Centers for Disease Control and Prevention, there are about 1 in 33 cases of birth defects in the United States, and almost 20% of all infant deaths can be attributed to them. This creates a sense of urgency to incorporate early prenatal genetic screening, thus fueling the requirement for sophisticated NIPT services in North America.
Asia Pacific market is expected to register the highest CAGR during the forecast period. This is due to the high birth rate and the rise in the number of people in the middle class, who are spending more on sophisticated prenatal care facilities. In this region, there is an increase in the availability of private maternity facilities and diagnostic laboratories.
- According to UNICEF, India experiences nearly 25 million births per annum, which is close to one-fifth of the total yearly births worldwide. This high number of births provides a large market base to be addressed with regard to the development of the NIPT market in the country.
Europe non-invasive prenatal tests market is driven by the established prenatal screening programs in Germany, France, and the UK. The public healthcare infrastructure is increasingly incorporating non-invasive prenatal tests in the care of pregnant women with high-risk pregnancies.
Latin America noninvasive prenatal testing market growth is fueled by expansion of private diagnostic laboratories in Brazil and Mexico. Rising maternal age and increasing focus on early fetal abnormality detection are increasing test growth.
Middle East and Africa noninvasive prenatal testing market is driven by healthcare infrastructure investment in the UAE and Saudi Arabia. Expansion of specialized diagnostic centers is improving access to prenatal genetic screening. South Africa is observing gradual adoption through private healthcare providers.
Top Key Players & Market Share Insights:
The non-invasive prenatal tests market is highly competitive, with established molecular diagnostics and genomic tests service providers offering advanced non-invasive prenatal tests in the domestic and international markets. Market participants are focusing on technological innovation in next-generation sequencing (NGS), portfolio expansion, strategic collaborations with hospitals and diagnostic laboratories, and geographic expansion to strengthen their market presence. Key participants in the market for Noninvasive Prenatal Testing include:
- Illumina, Inc. – US
- Natera, Inc.– US
- Hoffmann-La Roche Ltd.– Switzerland
- BGI Genomics Co., Ltd. – China
- Laboratory Corporation of America Holdings – US
- Quest Diagnostics Incorporated – US
- Eurofins LifeCodexx GmbH – Germany
- Berry Genomics Co., Ltd. – China
- Myriad Genetics, Inc. – US
- Agilent Technologies, Inc. – US
Recent Industry Developments:
- In August 2025, Natera introduced Fetal Focus, a new non-invasive prenatal test that detects genetic conditions that can be inherited by the fetus. Fetal Focus added to the Non-Invasive Prenatal Testing category by providing parents with safer screening options for their babies without the risks of invasive procedures.
- In October 2024, YourGene Health and Genetix introduced the first non-invasive prenatal testing (NIPT) service in Colombia. The service enabled expectant parents to have access to genetic screening for their babies without undergoing invasive procedures. The expansion of the service helped to advance non-invasive prenatal testing.
Noninvasive Prenatal Testing Market Report Insights:
| Report Attributes | Report Details |
|---|---|
| Study Timeline | 2019-2035 |
| Market Size in 2035 (USD Trillion) | USD 19.36 Billion |
| CAGR (2025-2035) | 11.69% |
| By Gestation Period |
|
| By Pregnancy Risk |
|
| By Method |
|
| By Technology |
|
| By Product |
|
| By Application |
|
| By End Use |
|
| By Region |
|
| Key Players |
|
| Report Coverage |
|
Key Questions Answered in the Report
How big is the noninvasive prenatal testing market? +
The noninvasive prenatal testing market sizeis estimated to reach over USD 19.36 Billion by 2035 from a value of USD 5.74 Billion in 2024 and is projected to grow by USD 6.41 Billion in 2025, growing at a CAGR of 11.69% from 2025 to 2035.
Which segmentation details are covered in the noninvasive prenatal testing report? +
The noninvasive prenatal testing report includes specific segmentation details for gestation period, pregnancy risk, method, technology, product, application, end-use, and regions.
Which is the fastest segment anticipated to impact the market growth? +
In the noninvasive prenatal testing market, the low-risk pregnancy segment is anticipated to register the fastest growth during the forecast period, driven by expanding universal screening recommendations and increasing physician confidence in test accuracy.
Who are the major players in the noninvasive prenatal testing market? +
The key participants in the Noninvasive Prenatal Testing marketare Illumina, Inc. (US), Natera, Inc. (US), F. Hoffmann-La Roche Ltd. (Switzerland), BGI Genomics Co., Ltd. (China), Laboratory Corporation of America Holdings (US), Quest Diagnostics Incorporated (US), Eurofins LifeCodexx GmbH (Germany), Berry Genomics Co., Ltd. (China), Myriad Genetics, Inc. (US), and Agilent Technologies, Inc. (US), among others.
What are the key trends in the noninvasive prenatal testing market? +
The noninvasive prenatal testing market is shaped by key trends including increasing maternal age globally, expansion of first-trimester screening programs and rising adoption of cell-free DNA testing.
