Home > > Medical Devices > > Ablation Devices Market Size, Growth, Analysis 2024 To 2031
Ablation Devices Market - Size, Share, Industry Trends, and Forecasts (2024 - 2031)
ID : CBI_1625 | Updated on : | Author : Yogesh K | Category : Medical Devices
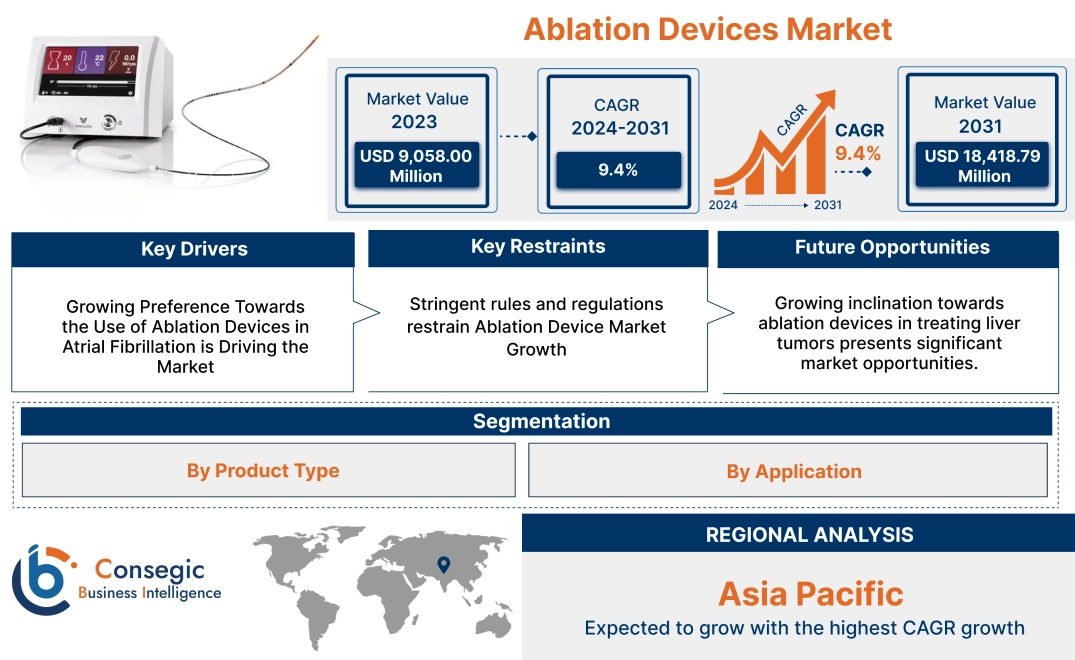
Ablation Devices Market Size:
Ablation Devices Market size is estimated to reach over USD 18,418.79 Million by 2031 from a value of USD 9,058.00 Million in 2023, growing at a CAGR of 9.4% from 2024 to 2031.
Ablation Devices Market Scope & Overview:
An ablation device is a medical device that uses various forms of energy such as radiofrequency, microwaves, and others to destroy tissue. These devices are used in ablation procedures in order to destroy abnormal tissue that is present in many conditions like atrial fibrillation, and tumors, among others. Laser ablation which is used to treat skin discolorations or lesions is one of the examples of ablation. There are different types of ablation devices available such as Radiofrequency Ablation, Cryoablation, Pulsed-field Ablation, and Microwave Ablation, among others. Benefits of this ablation device include fast recovery, immediate pain relief, efficiency, and others.
Ablation Devices Market Dynamics - (DRO) :
Key Drivers:
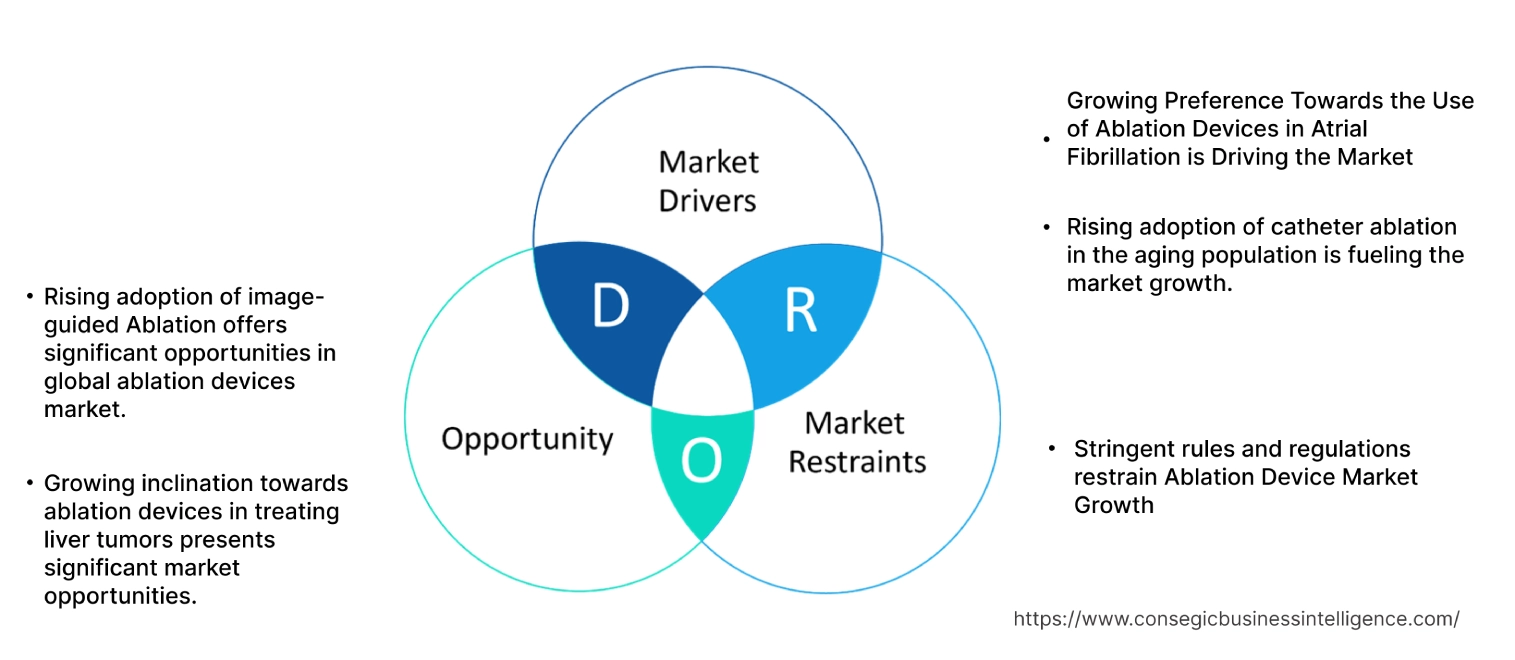
Growing Preference Towards the Use of Ablation Devices in Atrial Fibrillation is Driving the Market
Devices for ablation are minimally invasive devices used for treating atrial fibrillation. These devices are made up of different parts such as a generator unit, catheter, grounding pad, and connection cables. Radiofrequency ablation is one such type of ablation device that uses thermal energy to destroy small areas of tissue that are causing abnormal heartbeat in atrial fibrillation. While pulsed field devices use electrical pulses to cause nonthermal irreversible delivery of rapid, high-voltage pulsed electrical fields to tissue and induce cardiac cell death.
- According to the Journal of the American College of Cardiology, in 178 patients atrial fibrillation is treated with an ablation device with a focal Radio-Frequency/Pulse-Fielded catheter. It allows chronic lesion durability and good freedom for both paroxysmal and persistent atrial fibrillation.
Thus, devices for ablation are proven to be effective for both clinical as well as patient outcomes which is driving the ablation devices industry and ablation devices market growth.
Rising adoption of catheter ablation in the aging population is fueling the market growth.
The aging population is a major driver for the ablation devices market because as people age, they have a higher risk of complications from traditional surgical procedures due to age-related health issues. Ablation devices which offer minimally invasive treatment options, become particularly valuable for this demographic. The catheter Ablation is equipped with an electrode at its tip and inserted into the body with the help of an X-ray. It is proven to be a safer and more effective treatment in people aged more than 80 years for atrial fibrillation.
- In 2022, according to a research article in the European Heart Journal, Catheter ablation is used in patients aged ≥80 years with symptomatic paroxysmal, persistent, and long-standing Atrial Fibrillation. It shows favorable acute success and low complication rates in these patients.
In conclusion, as the global population continues to age, minimally invasive devices like ablation devices are growing significantly.
Key Restraints :
Stringent rules and regulations restrain Ablation Device Market Growth
Ablation devices are used in procedures, such as endometrial ablation, tumor ablation, pain management, and others. These devices are subject to stringent regulatory standards to ensure patient safety and efficacy. Government bodies like the European Union, classify medical devices into different classes, and according to that the manufacturers should adhere to rules and regulations.
- According to the European Union, the ablation devices are classified as class III presenting a high or medium potential for internal exposure. For implantable devices and class III devices, a premarket clinical investigation is compulsory.
Also for devices of ablation, the manufacturer must prepare a periodic safety update report for each device, and, where relevant, for each category or group of devices that should be updated annually.
These rules and regulations restrict market growth by increasing the time and expense needed to bring new devices to market.
Future Opportunities :
Growing inclination towards ablation devices in treating liver tumors presents significant market opportunities.
Ablation devices are used for tumor ablation which is defined as the direct application of chemical or thermal therapies to a tumor to destroy and eradicate tumor to obtain cellular necrosis either by thermal or freezing techniques. It includes radiofrequency ablation and microwave ablation which utilize thermocoagulation energy and microwave energy, respectively, to create a complete necrosis zone encircling the lesion in the liver tumor.
- According to the Journal of Surgery Open Science, thermal tumor ablation has gained popularity as an alternative therapy to liver resection for the treatment of malignant liver tumors secondary to its high efficacy and low morbidity
Thus, the growing inclination towards ablation in liver tumors presents significant ablation devices market opportunities due to the rising prevalence of liver cancer.
Rising adoption of image-guided Ablation offers significant opportunities in global ablation devices market.
The image-guided ablation includes integrating advanced imaging technologies such as Magnetic Resonance Imaging, Computer Tomography, and ultrasound in the ablation system. This image-guided ablation is equivalent to surgery to treat patients with early-stage non-small cell lung cancer or oligometastatic lung disease. Modalities available include radiofrequency ablation, microwave ablation, and cryoablation.
- According to a research article in Springer Open, Image-guided percutaneous lung ablation has proven to be a valid treatment alternative to surgery in patients with early-stage non-small cell lung carcinoma or oligometastatic lung disease. It exhibits excellent tolerance with very few complications.
Thus, image-guided ablation helps to provide safer treatments, effectively further strengthening the ablation devices market expansion.
Ablation Devices Market Segmental Analysis :
By Product Type:
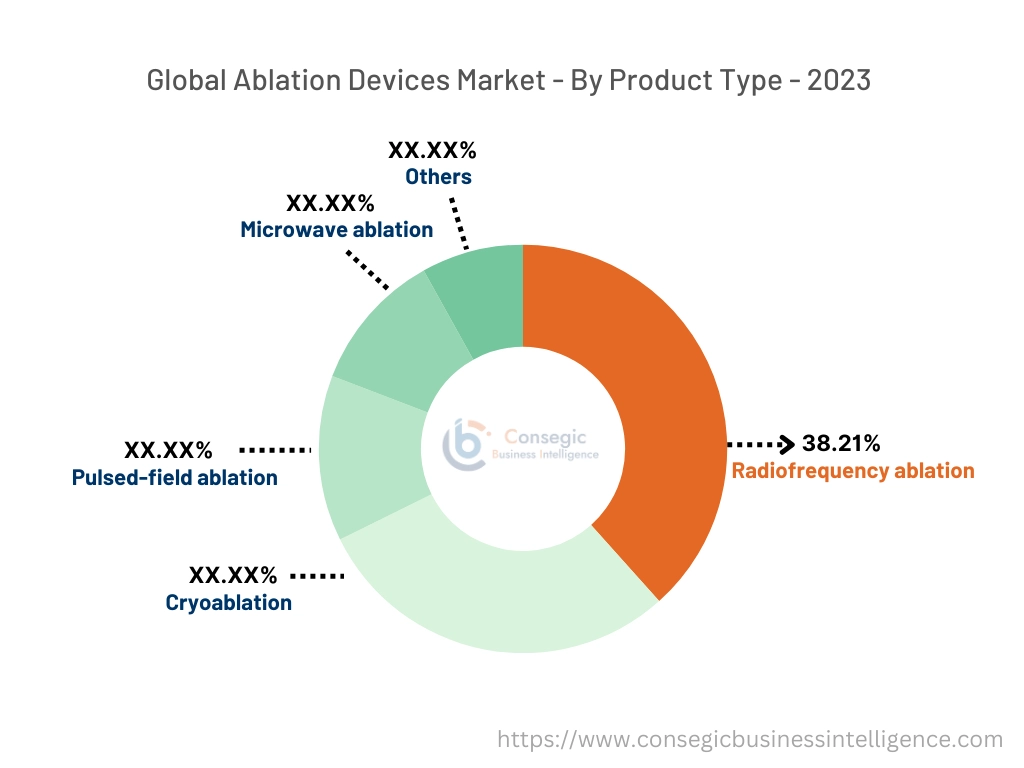
Based on the product type, the ablation devices market is divided into radiofrequency ablation, cryoablation, pulsed-field ablation, microwave ablation, and others.
Trends in Product Type:
- In the Ablation Devices Market trend, image-guided Radiofrequency Ablation improves treatment planning and minimizes damage to healthy tissues.
- Pulsed Field Ablation is a safer and faster ablation for the treatment of atrial fibrillation in the latest Ablation Devices Market trend.
Radiofrequency Ablation accounted for the largest revenue share of 38.21% in the year 2023.
Rational Protein Design is a strategy in protein engineering that involves making specific changes to a protein's amino acid sequence based on its known structure and function.
- Radiofrequency ablation devices incorporate a minimally invasive technique in which part of the electrical conduction system of the heart, tumor, sensory nerves, or dysfunctional tissue is ablated using the heat generated from medium frequency alternating current.
- It is used to treat a range of conditions, including benign and malignant tumors, chronic venous insufficiency in the legs, as well as chronic back and neck pain
- Radiofrequency ablation devices use an electrosurgical generator connected to bipolar electrode arrays to deliver thermal energy to tissue
- In July 2024, Stryker launched a MultiGen 2 Radiofrequency Generator for performing Radiofrequency ablation in chronic joint pain management. The MultiGen 2 Generator achieves target temperature faster, with fewer errors for increased reliability and efficiency
- Thus, Radiofrequency ablation is expected to maintain its leading position due to wider acceptability and efficiency as per trend and analysis
The Pulsed-Field Ablation in the product type is expected to grow at the fastest CAGR over the forecast period.
- Pulsed field ablation uses electrical pulses to cause nonthermal irreversible delivery of rapid, high-voltage pulsed electrical fields to tissue and induce cardiac cell death
- It is used most widely to treat tumors (cancer) and cardiac arrhythmias
- Pulsed field ablation is the latest ablation modality approved by the Food and Drug Administration that is used to restore a regular heartbeat
- In October 2024, Medtronic plc received United States Food and Drug Administration approval of the Affera™ Mapping and Ablation Device System with Sphere-9™ Catheter. This Sphere-9 catheter enables a single transeptal, zero-fluoroscopy, and zero-exchange workflow. It uniquely integrates both mapping and ablation technologies, offering the flexibility to use either pulsed field or radiofrequency energy devices.
- In summary, pulsed-field ablation is experiencing rapid growth due to government support and technological advancements according to analysis.
By Application:
Based on the application, the ablation devices market is divided into Cardiology, Oncology, Orthopedics, Pain Management, and others.
Trends in Application:
- In trend, ablation devices market demand for cardiac ablation devices is motivated by a rise in the prevalence of cardiovascular diseases like cardiac arrhythmia, atrial fibrillation, and stroke
- As per industry trends, the growing need for safer therapeutic choices and the rising incidence of cancer are two key drivers of the ablation devices market expansion in oncology
Cardiology accounts for the largest market share of the Ablation Devices Market by application.
- The ablation devices are used in cardiology for the treatment of atrial arrhythmias, atrial fibrillation, and others
- It uses thin, flexible tubes called catheters and heat or cold energy to create tiny scars in the heart
- These scars stop unusual electrical signals that move through the heart and cause an uneven heartbeat
- In July 2024, Pulse Biosciences received the Food and Drug Administration's breakthrough device designation for its new CellFX Nanosecond Pulsed Field Ablation system. This system is associated with a reduced risk of adverse events due to its ability to destroy heart cells causing arrhythmias without the use of extreme heat or cold in atrial fibrillation
- Thus, as per the analysis cardiology remains the leading application segment in ablation devices due to their efficiency in treating cardiology conditions
The Oncology in the application is expected to grow at the fastest CAGR over the forecast period.
- Ablation devices use alternating current to induce cell death in cancer. It includes a generator, grounding pads, and a needle electrode/catheter
- The needle is connected to the generator which creates an alternating current that travels between the needle and the grounding pads on the patient's thighs/legs. The alternating current creates ionic inner tension in the treatment zone, creating frictional heat that modifies the tumor
- Depending on the size of the tumor, ablation zones between 2 to 5 cm are created in 10 to 30 minutes
- In July 2024, Innoblative Designs announced that it began a first-in-human clinical experience for SIRA Radiofrequency ablation electrosurgical device for its breast cancer ablation system. SIRA uses radiofrequency ablation through a unique spherical shape that circumferentially delivers radiofrequency energy.
- Hence, the use of ablation devices in oncology is growing supported by innovative developments and improved outcomes according to current trends and analysis
Regional Analysis:
The regional segment includes North America, Europe, Asia Pacific, the Middle East and Africa, and Latin America.
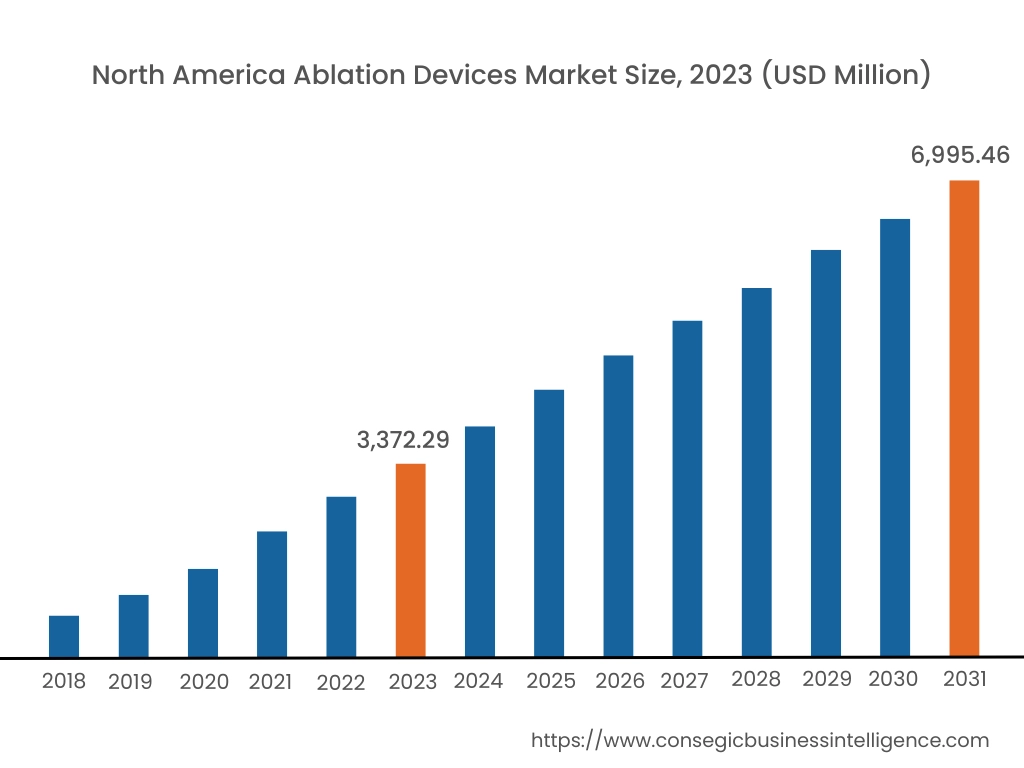
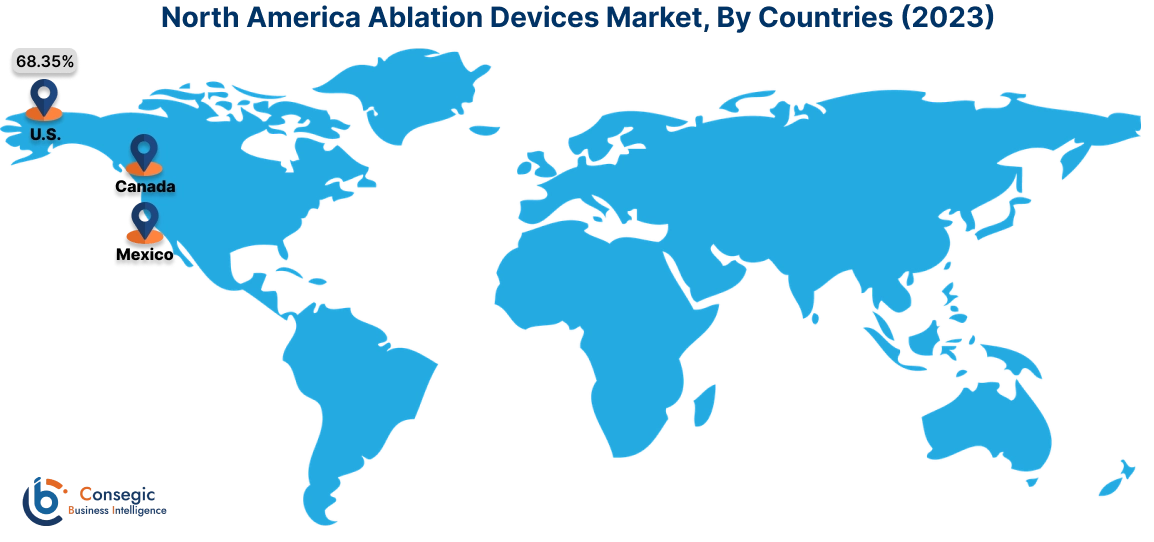
In 2023, North America accounted for the highest market share at 37.23% and was valued at USD 3,372.29 Million, and is expected to reach USD 6,995.46 Million in 2031. In North America, the U.S. accounted for the highest market share of 68.35% during the base year of 2023. North America holds a significant part in the global ablation device market driven by the high prevalence of cardiovascular diseases, cancer, and other chronic conditions that benefit from minimally invasive treatments. The advanced healthcare infrastructure, rising healthcare expenditure, and adoption of innovative technologies in the U.S. and Canada contribute to the region's expansion in ablation therapy.
- In December 2023, NeuroOne Medical Technologies Corporation announced that it had received US Food and Drug Administration (FDA) 510(k) clearance to market its OneRF ablation system. It includes ablation devices and other equipment for the creation of radiofrequency lesions in nervous tissue for functional neurosurgical procedures.
Thus, according to the ablation devices market analysis, North America is expected to dominate the global ablation device market supported by advanced healthcare infrastructure, government support, and rising use in healthcare procedures.
Asia-Pacific is expected to witness the fastest CAGR over the forecast period of 10.1% during 2024-2031. The Asia-Pacific region is experiencing rapid growth in the ablation devices market due to rising healthcare needs, particularly in populous countries like China, India, and Japan. Additionally, governments in Asia-Pacific are investing heavily in expanding healthcare infrastructure with improvements in access to healthcare services and rising healthcare spending.
- In November 2021, India Medtronic Private Limited announced the launch of the Arctic Front Cardiac Cryoablation Catheter System, a type of ablation device. It is the first and only cryoballoon catheter approved by the Central Drugs Standard Control Organization (CDSCO) for the treatment of atrial fibrillation in India. Cryoballoon ablation is a minimally invasive technique that offers a short recovery time and comparable safety and efficacy to standard radiofrequency therapy, with the benefit of greater treatment efficiency.
Therefore, analysis shows that the Asia-Pacific region is expected to grow faster, capitalizing on both demand and infrastructural advancements in the ablation devices market.
The European region's demand for ablation tools is experiencing significant growth driven by advancements in technology, a high incidence of cancer and cardiovascular diseases, and increasing investment in healthcare infrastructure. Key factors include a growing geriatric population, favorable reimbursement policies, and robust healthcare infrastructure in many parts of the region. Countries like Germany, France, and the United Kingdom are major contributors to market growth due to strong healthcare systems and ongoing technological advancements. The region is witnessing a surge in demand for radiofrequency and microwave ablation, particularly for oncology and cardiology applications. Additionally, ongoing research and development and the presence of major medical device manufacturers further boost market expansion.
The ablation devices market in the Middle East and Africa is at a nascent stage. The market is steadily growing, fueled by an increasing prevalence of chronic diseases such as cancer and cardiovascular disorders, coupled with rising awareness about minimally invasive treatments. In the Middle East, affluent countries like Saudi Arabia and the UAE are leading the market due to improved healthcare infrastructure, and growing investments in advanced healthcare technologies, while Africa is gradually improving healthcare access and infrastructure. Despite this demand for the market is expected to rise, economic disparities and limited healthcare facilities in some parts of the region continue to restrict the market
In Latin America, the ablation devices market demand is emerging with steady growth potential. While healthcare infrastructure varies across the region, improving economics, rising investment in healthcare, and increasing awareness of minimally invasive techniques drive market adoption in countries like Brazil and Mexico. The demand for minimally invasive techniques, including cardiac ablation for cardiac arrhythmia, and tumor ablation for oncology is contributing to the market expansion. Additionally, advancements in ablation technologies such as the introduction of pulsed fields and cryoblation devices are further boosting the market. The healthcare sector in this region is expected to see continued investment in ablation technologies, driven by the need for effective, less invasive treatment options.
Top Key Players & Market Share Insights:
The ablation devices market is highly competitive with major players providing products and services to the national and international markets. Key players are adopting several strategies in research and development (R&D), product innovation, and end-user launches to hold a strong position in the global ablation devices market. Key players in the ablation devices industry include -
- Abbott (United States)
- Boston Scientific Corporation (United States)
- Medtronic (United States)
- AngioDynamics (United States)
- EDAT TMS (France)
- Johnson & Johnson Services, Inc. (United States)
- Varian Medical Systems, Inc. (United States)
- Biotronik (United States)
- Hologic, Inc (United States)
- Avanos Medical, Inc (United States)
Recent Industry Developments :
Approvals and Patents:
- In November 2024, the U.S. Food & Drug Administration approved Johnson & Johnson's Varipulse device. The device, Varipulse, is a pulsed field ablation system that uses small burns or freezes to cause some scarring in the inside of the heart to help break up the electrical signals that cause irregular heartbeats.
- In 2023, Abbott received FDA Approval for a TactiFlex™ Ablation Catheter for Treatment of Abnormal Heart Rhythm. TactiFlex™ Ablation Catheter, Sensor Enabled™, is the world's first ablation catheter designed with a unique flexible electrode tip and contact force sensing to treat patients with atrial fibrillation. The TactiFlex catheter is designed to be used with Abbott's EnSite X EP System, an industry-leading heart mapping system, which allows physicians to view and precisely identify areas in the heart that require ablation.
- In 2024, British technology start-up Ablatus received a new UK patent for its ablation technology. It offers new minimally invasive treatment options for up to 12 million women in the UK with uterine fibroids.
Investments and Fundings:
- In 2024, Kardium raises $104M to develop a pulsed field ablation system. The funds will be used to complete Kardium's Pulsar clinical study needed to obtain regulatory approvals for its Globe mapping and ablation system.
Ablation Devices Market Report Insights :
| Report Attributes | Report Details |
| Study Timeline | 2018-2031 |
| Market Size in 2031 | USD 18,418.79 Million |
| CAGR (2024-2031) | 9.4% |
| By Product Type |
|
| By Application |
|
| By Region |
|
| Key Players |
|
| North America | U.S. Canada Mexico |
| Europe | U.K. Germany France Spain Italy Russia Benelux Rest of Europe |
| APAC | China South Korea Japan India Australia ASEAN Rest of Asia-Pacific |
| Middle East and Africa | GCC Turkey South Africa Rest of MEA |
| LATAM | Brazil Argentina Chile Rest of LATAM |
| Report Coverage |
|
Key Questions Answered in the Report
How big is the Ablation Devices Market? +
Ablation Devices Market size is estimated to reach over USD 18,418.79 Million by 2031 from a value of USD 9,058.00 Million in 2023, growing at a CAGR of 9.4% from 2024 to 2031.
What specific segmentation details are covered in the Ablation Devices Market report? +
The Ablation Devices Market report includes specific segmentation details for product type and application
Which is the fastest-growing region in the Ablation Devices Market? +
Asia Pacific is the fastest-growing region in the Ablation Devices Market
Who are the major players in the Ablation Devices Market? +
The key participants in the Ablation Devices market are Abbott (United States), Boston Scientific Corporation (United States), Johnson & Johnson Services Inc. (United States), Varian Medical Systems Inc. (United States), Biotronik (Germany), Medtronic (United States), AngioDynamics (United States), EDAT TMS (France), Hologic Inc (United States), Avanos Medica Inc (United States)